Preliminary Clinical Outcomes Following Biologic Augmentation of Arthroscopic Rotator Cuff Repair Using Subacromial Bursa, Concentrated Bone Marrow Aspirate, and Platelet-Rich Plasma
- PMID: 33376995
- PMCID: PMC7754610
- DOI: 10.1016/j.asmr.2020.07.019
Preliminary Clinical Outcomes Following Biologic Augmentation of Arthroscopic Rotator Cuff Repair Using Subacromial Bursa, Concentrated Bone Marrow Aspirate, and Platelet-Rich Plasma
Abstract
Purpose: To evaluate the clinical outcomes of patients who underwent arthroscopic rotator cuff repair augmented using subacromial bursa, concentrated bone marrow aspirate (cBMA), and platelet-rich plasma.
Methods: Sixteen patients were included in the study who underwent arthroscopic rotator cuff repair augmented using subacromial bursa, cBMA, and platelet-rich plasma from January 2018 to July 2018 and had a minimum 1-year follow-up. American Shoulder and Elbow Surgeons (ASES), Simple Shoulder Test, Constant-Murley, and Single Assessment Numerical Evaluation (SANE) scores were collected preoperatively and at terminal follow-up. To determine the clinical relevance of ASES scores, the minimal clinically important difference, substantial clinical benefit, and the patient acceptable symptomatic state thresholds were used. In vitro cellular proliferation of subacromial bursa (nucleated cells/gram) and cBMA (nucleated cells and colony-forming units/cc) samples was evaluated and correlated to clinical outcomes scores.
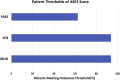
Results: Mean follow-up was 12.6 ± 1.8 months (range 12-19 months). Patients achieved significant improvement in ASES (45.8±22.5pre vs 88.5 ± 14.6post, Δ44.7 ± 20.7; P = .001), Simple Shoulder Test (4.3 ± 3.2pre vs 10.4 ± 1.6post, Δ5.7 ± 3.9, P = .002), Constant-Murley (44.3 ± 18.2pre vs 83.6 ± 17.5post, Δ37.2 ± 21.8; P = .001), SANE (13.3 ± 10.7pre vs 86.3 ± 17.5post, Δ71.9 ± 22.9; P = .001), and pain scores (5.0±2.8pre vs 1.1 ± 1.6post, Δ3.5±2.5, P = .001) at final follow-up. With regards to ASES score, 93.8% of patients achieved the minimal clinically important difference, 93.8% the substantial clinical benefit, and 62.5% reached or exceeded the patient acceptable symptomatic state criteria. There was a significant positive correlation of nucleated cell count of cBMA with postoperative SANE score (r = 0.707; P = .015) and delta in ASES score (r = 0.727; P = .011). All other correlations were found to be nonsignificant (P > .05, respectively).
Conclusions: Patients undergoing arthroscopic rotator cuff repair augmented using the Mega-Clot with bursa technique achieved significant improvement in functional outcomes at a minimum 1-year follow-up, with 93.8% of patients reaching substantial clinical benefit.
Level of evidence: Level IV, therapeutic case series.
© 2020 by the Arthroscopy Association of North America. Published by Elsevier.
Figures







Similar articles
-
Clinical Outcomes Following Biologically Enhanced Patch Augmentation Repair as a Salvage Procedure for Revision Massive Rotator Cuff Tears.Arthroscopy. 2020 Jun;36(6):1542-1551. doi: 10.1016/j.arthro.2020.02.006. Epub 2020 Mar 30. Arthroscopy. 2020. PMID: 32241704
-
Arthroscopic Rotator Cuff Repair with a Fibrin Scaffold Containing Growth Factors and Autologous Progenitor Cells Derived From Humeral cBMA Improves Clinical Outcomes in High Risk Patients.Arthrosc Sports Med Rehabil. 2022 Aug 1;4(5):e1629-e1637. doi: 10.1016/j.asmr.2022.06.006. eCollection 2022 Oct. Arthrosc Sports Med Rehabil. 2022. PMID: 36312725 Free PMC article.
-
Arthroscopic rotator cuff repair with biologically enhanced patch augmentation.Oper Orthop Traumatol. 2022 Feb;34(1):4-12. doi: 10.1007/s00064-021-00754-3. Epub 2021 Dec 10. Oper Orthop Traumatol. 2022. PMID: 34890027 Review. English.
-
Prospective Randomized Trial of Biologic Augmentation With Bone Marrow Aspirate Concentrate in Patients Undergoing Arthroscopic Rotator Cuff Repair.Am J Sports Med. 2023 Apr;51(5):1234-1242. doi: 10.1177/03635465231154601. Epub 2023 Feb 22. Am J Sports Med. 2023. PMID: 36811557 Clinical Trial.
-
Platelet-Rich Plasma Augmentation of Arthroscopic Rotator Cuff Repair Lowers Retear Rates and Improves Short-Term Postoperative Functional Outcome Scores: A Systematic Review of Meta-Analyses.Arthrosc Sports Med Rehabil. 2022 Feb 5;4(2):e823-e833. doi: 10.1016/j.asmr.2021.12.012. eCollection 2022 Apr. Arthrosc Sports Med Rehabil. 2022. PMID: 35494273 Free PMC article. Review.
Cited by
-
Combined Biologic Augmentation Strategies with Collagen Patch Graft, Microfractures, and Platelet Concentrate Injections Improve Functional and Structural Outcomes of Arthroscopic Revision Rotator Cuff Repair.J Clin Med. 2023 Sep 1;12(17):5694. doi: 10.3390/jcm12175694. J Clin Med. 2023. PMID: 37685760 Free PMC article.
-
The role of injections of mesenchymal stem cells as an augmentation tool in rotator cuff repair: a systematic review.JSES Rev Rep Tech. 2025 Jan 13;5(2):231-242. doi: 10.1016/j.xrrt.2024.12.003. eCollection 2025 May. JSES Rev Rep Tech. 2025. PMID: 40321851 Free PMC article. Review.
-
Arthroscopic Biological Augmentation With Subacromial Bursa for Bursal-Sided Partial-Thickness Rotator Cuff Tears.Orthop J Sports Med. 2023 Aug 29;11(8):23259671231190335. doi: 10.1177/23259671231190335. eCollection 2023 Aug. Orthop J Sports Med. 2023. PMID: 37655250 Free PMC article.
-
Clinical perspectives for repairing rotator cuff injuries with multi-tissue regenerative approaches.J Orthop Translat. 2022 Aug 24;36:91-108. doi: 10.1016/j.jot.2022.06.004. eCollection 2022 Sep. J Orthop Translat. 2022. PMID: 36090820 Free PMC article. Review.
-
A Comparison of Functional Outcomes in Rotator Cuff Repairs Using Adjunctive Bone Marrow Aspirate Concentrate vs. Bone Marrow Aspirate Concentrate With Platelet-Rich Plasma: A Systematic Review and Meta-Analysis.Cureus. 2024 Aug 23;16(8):e67594. doi: 10.7759/cureus.67594. eCollection 2024 Aug. Cureus. 2024. PMID: 39310448 Free PMC article. Review.
References
-
- Galatz L., Ball C., Teefey S. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am. 2004;86:219–224. - PubMed
-
- Cummins C.A., Appleyard R.C., Strickland S., Haen P.S., Chen S., Murrell G.A. Rotator cuff repair: an ex vivo analysis of suture anchor repair techniques on initial load to failure. Arthroscopy. 2005;21:1236–1241. - PubMed
-
- Cole B.J., ElAttrache N.S., Anbari A. Arthroscopic rotator cuff repairs: an anatomic and biomechanical rationale for different suture-anchor repair configurations. Arthroscopy. 2007;23:662–669. - PubMed
-
- Gamradt S.C., Gallo R.A., Adler R.S., et al. Vascularity of the supraspinatus tendon three months after repair: Characterization using contrast-enhanced ultrasound. J Shoulder Elbow Surg. 2010;19:73–80. - PubMed
-
- Zumstein M.A., Rumian A., Thelu C.E., et al. SECEC Research Grant 2008 II: Use of platelet- and leucocyte-rich fibrin (L-PRF) does not affect late rotator cuff tendon healing: A prospective randomized controlled study. J Shoulder Elbow Surg. 2016;25:2–11. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

