Cost per cumulative clinical benefit of biologic therapies for patients with plaque psoriasis: a systematic review
- PMID: 33377444
- PMCID: PMC10391184
- DOI: 10.18553/jmcp.2021.27.1.084
Cost per cumulative clinical benefit of biologic therapies for patients with plaque psoriasis: a systematic review
Abstract
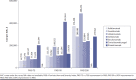
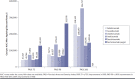
BACKGROUND: Measuring cumulative clinical treatment benefit over time captures speed and magnitude of effects. Assessing the cost of biologics relative to their cumulative clinical benefits versus a single time point represents an alternative to evaluate the value of a given biologic used to treat psoriasis. OBJECTIVE: To compare cumulative benefit and cost per cumulative benefit of biologics in treatment of moderate to severe psoriasis from a network meta-analysis (NMA). METHODS: Biologics included in the analysis were ixekizumab, adalimumab, guselkumab, ustekinumab, secukinumab, risankizumab, and certolizumab pegol. Psoriasis Area and Severity Index (PASI) responses over the initial 16-week treatment period were obtained from 31 articles. Cumulative benefits for PASI 75, PASI 90, and PASI 100 responses were measured as area under the curve (AUC) using the trapezoidal method. Bayesian-based NMA modeled percent maximum AUC through week 16 (%Max_AUCW16). The AUC estimates over 16 weeks were converted to total skin clearance threshold days achieved for PASI 75, PASI 90, and PASI 100 with each biologic. Cost per cumulative benefit was estimated by multiplying number of doses (per FDA label) by nationally representative discounted wholesale acquisition costs (WACs) for 16 weeks of treatment divided by %Max_AUCW16. The primary cost analysis used WACs, including week 16 doses. Co-primary cost analysis used discounted WACs, including week 16 doses. Sensitivity analysis was conducted using WACs and discounted WACs, excluding doses administered at week 16. RESULTS: Among biologics with available week 16 AUC data for PASI 90 and PASI 100, cumulative benefits over the initial 16-week treatment period ranged from 20.2% (certolizumab pegol) to 47.0% (ixekizumab) for PASI 90 and from 7.4% (adalimumab) to 22.2% (ixekizumab) for PASI 100. The total number of estimated PASI 90 and PASI 100 days achieved over the first 16 weeks of treatment was highest with ixekizumab (53 days and 25 days, respectively). In the primary analysis, guselkumab had the lowest cost per cumulative benefit (95% credible interval [CrI]; $99,742 [$89,941-$111,653]), followed by ixekizumab ($108,906 [$95,928-$126,093]) and adalimumab ($111,233 [$97,549-$129,022]) for PASI 90, and ixekizumab had the lowest cost per cumulative benefit ($230,884 [$191,611-$291,115]), followed by secukinumab ($238,945 [$204,029-$288,072]) and risankizumab ($279,968 [$250,683-$316,872]) for PASI 100 responses. In the co-primary analysis, ixekizumab had the lowest discounted cost per AUC (95% CrI; $60,988 [$53,719-$70,612]), followed by guselkumab ($66,827 [$60,260-$74,807]) and secukinumab ($69,622 [$61,783-$79,786]) for PASI 90, and ixekizumab had the lowest cost per cumulative benefit ($129,295 [$107,302-$163,024]), followed by secukinumab ($148,146 [$126,498-$178,605]) and guselkumab ($188,190 [$166,791-$215,969]) for PASI 100 responses. Conclusions: Among biologics studied, ixekizumab demonstrated the greatest cumulative clinical benefit, maintaining the lowest cost per cumulative benefit for PASI 100 responses and lowest discounted cost per cumulative benefit for PASI 90 and PASI 100 responses for moderate to severe psoriasis over the initial 16-week treatment period. DISCLOSURES: This study was funded by Eli Lilly and Company (Indianapolis, IN). Blauvelt has served as a scientific adviser and/or clinical study investigator for AbbVie, Aclaris, Almirall, Arena, Athenex, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant, Dermira, Eli Lilly and Company, Forte, Galderma, Incyte, Janssen, Leo, Novartis, Ortho, Pfizer, Rapt, Regeneron, Sandoz, Sanofi Genzyme, Sun Pharma, and UCB Pharma and as a paid speaker for AbbVie. Burge, Zhu, Malatestinic, Brnabic, Guo, and Janardhanan are employees and shareholder of Eli Lilly and Company.
Conflict of interest statement
This study was funded by Eli Lilly and Company (Indianapolis, IN). Blauvelt has served as a scientific adviser and/or clinical study investigator for AbbVie, Aclaris, Almirall, Arena, Athenex, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant, Dermira, Eli Lilly and Company, Forte, Galderma, Incyte, Janssen, Leo, Novartis, Ortho, Pfizer, Rapt, Regeneron, Sandoz, Sanofi Genzyme, Sun Pharma, and UCB Pharma and as a paid speaker for AbbVie. Burge, Zhu, Malatestinic, Brnabic, Guo, and Janardhanan are employees and shareholder of Eli Lilly and Company.
Figures
References
-
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70(3):512-16. - PubMed
-
- Puig L. PASI90 response: the new standard in therapeutic efficacy for psoriasis. J Eur Acad Dermatol Venereol. 2015;29(4):645-48. - PubMed
-
- Gordon KB, Blauvelt A, Papp KA, et al. Phase 3 trials of ixekizumab in moderate-to-severe plaque psoriasis. N Engl J Med. 2016;375(4):345-56. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical