The Interaction Between the Ventrolateral Preoptic Nucleus and the Tuberomammillary Nucleus in Regulating the Sleep-Wakefulness Cycle
- PMID: 33381012
- PMCID: PMC7767984
- DOI: 10.3389/fnins.2020.615854
The Interaction Between the Ventrolateral Preoptic Nucleus and the Tuberomammillary Nucleus in Regulating the Sleep-Wakefulness Cycle
Abstract
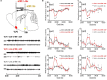
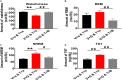
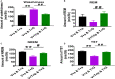
The ventrolateral preoptic nucleus (VLPO) in the anterior hypothalamus and the tuberomammillary nucleus (TMN) in the posterior hypothalamus are critical regions which involve the regulation of sleep-wakefulness flip-flop in the central nervous system. Most of the VLPO neurons are sleep-promoting neurons, which co-express γ-aminobutyric acid (GABA) and galanin, while TMN neurons express histamine (HA), a key wake-promoting neurotransmitter. Previous studies have shown that the two regions are innervated between each other, but how to regulate the sleep-wake cycle are not yet clear. Here, bicuculline (Bic), a GABA A -receptor antagonist, L-glutamate (L-Glu), an excitatory neurotransmitter, and triprolidine (Trip), a HA1 receptor (HRH1) inhibitor, were bilaterally microinjected into TMN or VLPO after surgically implanting the electroencephalogram (EEG) and electromyography (EMG) electrode recording system. Microinjecting L-Glu into VLPO during the night significantly increased the NREM sleep time, and this phenomenon was weakened after selectively blocking GABA A receptors with Bic microinjected into TMN. Those results reveal that VLPO neurons activated, which may inhibit TMN neurons inducing sleep via GABA A receptors. On the contrary, exciting TMN neurons by L-Glu during the day, the wakefulness time was significantly increased. These phenomena were reversed by blocking HRH1 with Trip microinjected into VLPO. Those results reveal that TMN neuron activating may manipulate VLPO neurons via HRH1, and induce wakefulness. In conclusion, VLPO GABAergic neurons and TMN histaminergic neurons may interact with each other in regulating the sleep-wake cycle.
Keywords: GABAA-receptor; HRH1; L-glutamate; TMN; VLPO; bicuculline; sleep-wake circuitry.
Copyright © 2020 Cheng, Wu, Zhang, Ding, Fan, Chen, Zhang and Wang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Optogenetic-mediated release of histamine reveals distal and autoregulatory mechanisms for controlling arousal.J Neurosci. 2014 Apr 23;34(17):6023-9. doi: 10.1523/JNEUROSCI.4838-13.2014. J Neurosci. 2014. PMID: 24760861 Free PMC article.
-
Innervation of histaminergic tuberomammillary neurons by GABAergic and galaninergic neurons in the ventrolateral preoptic nucleus of the rat.J Neurosci. 1998 Jun 15;18(12):4705-21. doi: 10.1523/JNEUROSCI.18-12-04705.1998. J Neurosci. 1998. PMID: 9614245 Free PMC article.
-
Monoamines Inhibit GABAergic Neurons in Ventrolateral Preoptic Area That Make Direct Synaptic Connections to Hypothalamic Arousal Neurons.J Neurosci. 2018 Jul 11;38(28):6366-6378. doi: 10.1523/JNEUROSCI.2835-17.2018. Epub 2018 Jun 18. J Neurosci. 2018. PMID: 29915137 Free PMC article.
-
[Sleep-wake regulation by prostaglandin D2 and adenosine].Brain Nerve. 2012 Jun;64(6):621-8. Brain Nerve. 2012. PMID: 22647469 Review. Japanese.
-
Sleep circuitry and the hypnotic mechanism of GABAA drugs.J Clin Sleep Med. 2006 Apr 15;2(2):S19-26. J Clin Sleep Med. 2006. PMID: 17557503 Review.
Cited by
-
Tfap2b acts in GABAergic neurons to control sleep in mice.Sci Rep. 2023 May 17;13(1):8026. doi: 10.1038/s41598-023-34772-x. Sci Rep. 2023. PMID: 37198238 Free PMC article.
-
Hypothalamic GABAergic neurocircuitry in the regulation of energy homeostasis and sleep/wake control.Med Rev (2021). 2022 Sep 14;2(5):531-540. doi: 10.1515/mr-2022-0022. eCollection 2022 Oct. Med Rev (2021). 2022. PMID: 37724165 Free PMC article. Review.
-
Noradrenergic tone is not required for neuronal activity-induced rebound sleep in zebrafish.J Comp Physiol B. 2024 Jun;194(3):279-298. doi: 10.1007/s00360-023-01504-6. Epub 2023 Jul 22. J Comp Physiol B. 2024. PMID: 37480493 Free PMC article.
-
The α2 Adrenoceptor Agonist and Sedative/Anaesthetic Dexmedetomidine Excites Diverse Neuronal Types in the Ventrolateral Preoptic Area of Male Mice.ASN Neuro. 2023 Jan-Dec;15:17590914231191016. doi: 10.1177/17590914231191016. ASN Neuro. 2023. PMID: 37499170 Free PMC article.
-
Clinical Spectrum and Trajectory of Innovative Therapeutic Interventions for Insomnia: A Perspective.Aging Dis. 2023 Aug 1;14(4):1038-1069. doi: 10.14336/AD.2022.1203. Aging Dis. 2023. PMID: 37163444 Free PMC article.
References
-
- Borbely A. A. (1982). A two process model of sleep regulation. Hum. Neurobiol. 1 195–204. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

