Molecular Modulation of Fetal Liver Hematopoietic Stem Cell Mobilization into Fetal Bone Marrow in Mice
- PMID: 33381191
- PMCID: PMC7755487
- DOI: 10.1155/2020/8885154
Molecular Modulation of Fetal Liver Hematopoietic Stem Cell Mobilization into Fetal Bone Marrow in Mice
Abstract
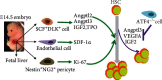
Development of hematopoietic stem cells is a complex process, which has been extensively investigated. Hematopoietic stem cells (HSCs) in mouse fetal liver are highly expanded to prepare for mobilization of HSCs into the fetal bone marrow. It is not completely known how the fetal liver niche regulates HSC expansion without loss of self-renewal ability. We reviewed current progress about the effects of fetal liver niche, chemokine, cytokine, and signaling pathways on HSC self-renewal, proliferation, and expansion. We discussed the molecular regulations of fetal HSC expansion in mouse and zebrafish. It is also unknown how HSCs from the fetal liver mobilize, circulate, and reside into the fetal bone marrow niche. We reviewed how extrinsic and intrinsic factors regulate mobilization of fetal liver HSCs into the fetal bone marrow, which provides tools to improve HSC engraftment efficiency during HSC transplantation. Understanding the regulation of fetal liver HSC mobilization into the fetal bone marrow will help us to design proper clinical therapeutic protocol for disease treatment like leukemia during pregnancy. We prospect that fetal cells, including hepatocytes and endothelial and hematopoietic cells, might regulate fetal liver HSC expansion. Components from vascular endothelial cells and bones might also modulate the lodging of fetal liver HSCs into the bone marrow. The current review holds great potential to deeply understand the molecular regulations of HSCs in the fetal liver and bone marrow in mammals, which will be helpful to efficiently expand HSCs in vitro.
Copyright © 2020 Huihong Zeng et al.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
Temporal modulation of calcium sensing in hematopoietic stem cells is crucial for proper stem cell expansion and engraftment.J Cell Physiol. 2020 Dec;235(12):9644-9666. doi: 10.1002/jcp.29777. Epub 2020 May 11. J Cell Physiol. 2020. PMID: 32394484
-
Mobilization and homing of hematopoietic stem cells.Adv Exp Med Biol. 2012;741:152-70. doi: 10.1007/978-1-4614-2098-9_11. Adv Exp Med Biol. 2012. PMID: 22457109 Review.
-
Current approaches in biomaterial-based hematopoietic stem cell niches.Acta Biomater. 2018 May;72:1-15. doi: 10.1016/j.actbio.2018.03.028. Epub 2018 Mar 22. Acta Biomater. 2018. PMID: 29578087 Review.
-
Circulation and chemotaxis of fetal hematopoietic stem cells.PLoS Biol. 2004 Mar;2(3):E75. doi: 10.1371/journal.pbio.0020075. Epub 2004 Mar 16. PLoS Biol. 2004. PMID: 15024423 Free PMC article.
-
Crosstalk Between the Hepatic and Hematopoietic Systems During Embryonic Development.Front Cell Dev Biol. 2020 Jul 22;8:612. doi: 10.3389/fcell.2020.00612. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32793589 Free PMC article. Review.
Cited by
-
Maternal NO2 exposure and fetal growth restriction: Hypoxia transmission and lncRNAs-proinflammation-mediated abnormal hematopoiesis.Proc Natl Acad Sci U S A. 2024 Oct 29;121(44):e2409597121. doi: 10.1073/pnas.2409597121. Epub 2024 Oct 21. Proc Natl Acad Sci U S A. 2024. PMID: 39432779 Free PMC article.
References
-
- Chatterjee R., Chattopadhyay S., Law S. Alteration of classical and hematopoiesis specific p53 pathway in the bone marrow hematopoietic stem/progenitor compartment facilitates leukemia progression in experimental mice. Leukemia Research. 2016;47:70–77. doi: 10.1016/j.leukres.2016.05.014. - DOI - PubMed
-
- Kita K., Xiu F., Jeschke M. G. Ex vivoexpansion of hematopoietic stem and progenitor cells: Recent advances. World Journal of Hematology. 2014;3(2):p. 18. doi: 10.5315/wjh.v3.i2.18. - DOI
Publication types
LinkOut - more resources
Full Text Sources

