Pomegranate peel extract ameliorates the severity of experimental autoimmune encephalomyelitis via modulation of gut microbiota
- PMID: 33382357
- PMCID: PMC7751635
- DOI: 10.1080/19490976.2020.1857515
Pomegranate peel extract ameliorates the severity of experimental autoimmune encephalomyelitis via modulation of gut microbiota
Abstract
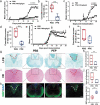
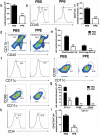
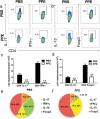
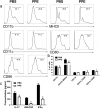
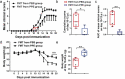
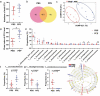
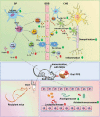
Multiple sclerosis (MS) is a CNS autoimmune disease characterized by demyelination and inflammatory infiltration with a high disability rate. Increasing evidence has demonstrated the importance of gut microbiota as an environmental risk factor in MS and its animal model experimental autoimmune encephalomyelitis (EAE). Diet is the main determinant of gut microbiota composition and function, which greatly affects the shaping of microbial structure. Pomegranate peel, a waste product in the production of juice, is rich in health-promoting compounds. However, its individual constituents, immunoregulatory activities, and action associated with bacterial diversity in the gut microbiota are largely unknown. Here, the main nutrient ingredients of pomegranate peel extract (PPE) were identified as phenols, flavonoids, amino acids, carbohydrates, fatty acids, lipids, nucleotides, organic acids, alcohols, and vitamins via metabolomics evaluation. We found, for the first time, oral PPE (100 mg/kg/day) not only effectively relieves EAE, inhibits CNS inflammatory factor infiltration and myelin loss, but also reshapes gut microbiota. Furthermore, recipient EAE mice with fecal transplantation from the PPE-treated donor delayed the disease development significantly. 16S rRNA gene sequencing revealed the increased gut microbiota richness in PPE-treated group. Among them, Lactobacillaceae enriched significantly, while Alcaligenaceae and Acidaminococcacea decreased remarkably. In conclusion, our data demonstrated that gut microbiota mediated the beneficial effects of oral PPE on EAE, and provided new ideas for developing the prebiotic value of pomegranate peel for the treatment of autoimmune diseases.
Keywords: 16s rRNA gene sequencing; experimental autoimmune encephalomyelitis; gut microbiota; multiple sclerosis; pomegranate peel extract.
Figures
Similar articles
-
Prebiotic inulin controls Th17 cells mediated central nervous system autoimmunity through modulating the gut microbiota and short chain fatty acids.Gut Microbes. 2024 Jan-Dec;16(1):2402547. doi: 10.1080/19490976.2024.2402547. Epub 2024 Sep 17. Gut Microbes. 2024. PMID: 39287045 Free PMC article.
-
Dietary ellagic acid therapy for CNS autoimmunity: Targeting on Alloprevotella rava and propionate metabolism.Microbiome. 2024 Jun 24;12(1):114. doi: 10.1186/s40168-024-01819-8. Microbiome. 2024. PMID: 38915127 Free PMC article.
-
Combination of cannabinoids, delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD), mitigates experimental autoimmune encephalomyelitis (EAE) by altering the gut microbiome.Brain Behav Immun. 2019 Nov;82:25-35. doi: 10.1016/j.bbi.2019.07.028. Epub 2019 Jul 26. Brain Behav Immun. 2019. PMID: 31356922 Free PMC article.
-
Gut Microbiota in Multiple Sclerosis and Experimental Autoimmune Encephalomyelitis: Current Applications and Future Perspectives.Mediators Inflamm. 2018 Apr 2;2018:8168717. doi: 10.1155/2018/8168717. eCollection 2018. Mediators Inflamm. 2018. PMID: 29805314 Free PMC article. Review.
-
Short-chain fatty acids and gut microbiota in multiple sclerosis.Acta Neurol Scand. 2019 Mar;139(3):208-219. doi: 10.1111/ane.13045. Epub 2018 Dec 3. Acta Neurol Scand. 2019. PMID: 30427062
Cited by
-
Combination of Chemically Characterized Pomegranate Extract and Hydrophilic Vitamins against Prolonged Fatigue: A Monocentric, Randomized, Double-Blind, Placebo-Controlled Clinical Trial.Nutrients. 2023 Jun 26;15(13):2883. doi: 10.3390/nu15132883. Nutrients. 2023. PMID: 37447210 Free PMC article. Clinical Trial.
-
The Effects of Different Feeding Regimes on Body Composition, Gut Microbial Population, and Susceptibility to Pathogenic Infection in Largemouth Bass.Microorganisms. 2023 May 22;11(5):1356. doi: 10.3390/microorganisms11051356. Microorganisms. 2023. PMID: 37317330 Free PMC article.
-
Migration effects on the intestinal microbiota of Tibetans.PeerJ. 2021 Oct 18;9:e12036. doi: 10.7717/peerj.12036. eCollection 2021. PeerJ. 2021. PMID: 34721954 Free PMC article.
-
Lycopene Supplemented Mediterranean Diet Ameliorates Experimental Autoimmune Encephalomyelitis (EAE) in Mice and Changes Intestinal Microbiome.J Neuroimmune Pharmacol. 2025 May 5;20(1):50. doi: 10.1007/s11481-025-10212-7. J Neuroimmune Pharmacol. 2025. PMID: 40323426 Free PMC article.
-
Pomegranate Extract Administration Reverses Loss of Motor Coordination and Prevents Oxidative Stress in Cerebellum of Aging Mice.Antioxidants (Basel). 2023 Nov 11;12(11):1991. doi: 10.3390/antiox12111991. Antioxidants (Basel). 2023. PMID: 38001844 Free PMC article.
References
-
- Correale J, Gaitan MI, Ysrraelit MC, Fiol MP. Progressive multiple sclerosis: from pathogenic mechanisms to treatment. Brain. 2017;140:527–546. - PubMed
-
- Lombardi VC, De Meirleir KL, Subramanian K, Nourani SM, Dagda RK, Delaney SL, Palotás A. Nutritional modulation of the intestinal microbiota; future opportunities for the prevention and treatment of neuroimmune and neuroinflammatory disease. J Nutr Biochem. 2018;61:1–16. doi:10.1016/j.jnutbio.2018.04.004. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
