Chloroquine and Sulfadoxine Derivatives Inhibit ZIKV Replication in Cervical Cells
- PMID: 33383619
- PMCID: PMC7823661
- DOI: 10.3390/v13010036
Chloroquine and Sulfadoxine Derivatives Inhibit ZIKV Replication in Cervical Cells
Abstract
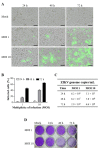
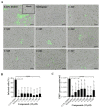
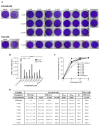
Despite the severe morbidity caused by Zika fever, its specific treatment is still a challenge for public health. Several research groups have investigated the drug repurposing of chloroquine. However, the highly toxic side effect induced by chloroquine paves the way for the improvement of this drug for use in Zika fever clinics. Our aim is to evaluate the anti-Zika virus (ZIKV) effect of hybrid compounds derived from chloroquine and sulfadoxine antimalarial drugs. The antiviral activity of hybrid compounds (C-Sd1 to C-Sd7) was assessed in an in-vitro model of human cervical and Vero cell lines infected with a Brazilian (BR) ZIKV strain. First, we evaluated the cytotoxic effect on cultures treated with up to 200 µM of C-Sds and observed CC50 values that ranged from 112.0 ± 1.8 to >200 µM in cervical cells and 43.2 ± 0.4 to 143.0 ± 1.3 µM in Vero cells. Then, the cultures were ZIKV-infected and treated with up to 25 µM of C-Sds for 48 h. The treatment of cervical cells with C-Sds at 12 µM induced a reduction of 79.8% ± 4.2% to 90.7% ± 1.5% of ZIKV-envelope glycoprotein expression in infected cells as compared to 36.8% ± 2.9% of infection in vehicle control. The viral load was also investigated and revealed a reduction of 2- to 3-logs of ZIKV genome copies/mL in culture supernatants compared to 6.7 ± 0.7 × 108 copies/mL in vehicle control. The dose-response curve by plaque-forming reduction (PFR) in cervical cells revealed a potent dose-dependent activity of C-Sds in inhibiting ZIKV replication, with PFR above 50% and 90% at 6 and 12 µM, respectively, while 25 µM inhibited 100% of viral progeny. The treatment of Vero cells at 12 µM led to 100% PFR, confirming the C-Sds activity in another cell type. Regarding effective concentration in cervical cells, the EC50 values ranged from 3.2 ± 0.1 to 5.0 ± 0.2 µM, and the EC90 values ranged from 7.2 ± 0.1 to 11.6 ± 0.1 µM, with selectivity index above 40 for most C-Sds, showing a good therapeutic window. Here, our aim is to investigate the anti-ZIKV activity of new hybrid compounds that show highly potent efficacy as inhibitors of ZIKV in-vitro infection. However, further studies will be needed to investigate whether these new chemical structures can lead to the improvement of chloroquine antiviral activity.
Keywords: Zika virus; antiviral effect; chloroquine; human cervical cells; hybrid compounds; sulfadoxine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Chloroquine, an Endocytosis Blocking Agent, Inhibits Zika Virus Infection in Different Cell Models.Viruses. 2016 Nov 29;8(12):322. doi: 10.3390/v8120322. Viruses. 2016. PMID: 27916837 Free PMC article.
-
Resveratrol affects Zika virus replication in vitro.Sci Rep. 2019 Oct 4;9(1):14336. doi: 10.1038/s41598-019-50674-3. Sci Rep. 2019. PMID: 31586088 Free PMC article.
-
The thiopurine nucleoside analogue 6-methylmercaptopurine riboside (6MMPr) effectively blocks Zika virus replication.Int J Antimicrob Agents. 2017 Dec;50(6):718-725. doi: 10.1016/j.ijantimicag.2017.08.016. Epub 2017 Aug 10. Int J Antimicrob Agents. 2017. PMID: 28803932
-
NS2B-NS3 protease inhibitors as promising compounds in the development of antivirals against Zika virus: A systematic review.J Med Virol. 2022 Feb;94(2):442-453. doi: 10.1002/jmv.27386. Epub 2021 Oct 20. J Med Virol. 2022. PMID: 34636434
-
Autophagy in Zika Virus Infection: A Possible Therapeutic Target to Counteract Viral Replication.Int J Mol Sci. 2019 Feb 28;20(5):1048. doi: 10.3390/ijms20051048. Int J Mol Sci. 2019. PMID: 30823365 Free PMC article. Review.
Cited by
-
Antiviral activity of chrysin and naringenin against porcine epidemic diarrhea virus infection.Front Vet Sci. 2023 Dec 7;10:1278997. doi: 10.3389/fvets.2023.1278997. eCollection 2023. Front Vet Sci. 2023. PMID: 38130439 Free PMC article.
-
Drugs that offer the potential to reduce hospitalization and mortality from SARS-CoV-2 infection: The possible role of the sigma-1 receptor and autophagy.Expert Opin Ther Targets. 2021 Jun;25(6):435-449. doi: 10.1080/14728222.2021.1952987. Epub 2021 Jul 15. Expert Opin Ther Targets. 2021. PMID: 34236922 Free PMC article. Review.
-
Discovery and structure-activity relationship study of novel isoxazole-based small molecules targeting Zika virus infections.RSC Med Chem. 2024 Jul 22;15(8):2792-2805. doi: 10.1039/d4md00240g. eCollection 2024 Aug 14. RSC Med Chem. 2024. PMID: 39157190 Free PMC article.
References
-
- Pan American Health Organization/World Health Organization Zika Cumulative Cases. [(accessed on 29 October 2020)]; Available online: https://www.paho.org/hq/index.php?option=com_content&view=article&id=123....
-
- Rozé B., Najioullah F., Fergé J.-L., Dorléans F., Apetse K., Barnay J.-L., Daudens-Vaysse E., Brouste Y., Césaire R., Fagour L., et al. Guillain-Barré Syndrome Associated With Zika Virus Infection in Martinique in 2016: A Prospective Study. Clin. Infect. Dis. 2017;65:1462–1468. doi: 10.1093/cid/cix588. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

