Androgen Reduces Mitochondrial Respiration in Mouse Brown Adipocytes: A Model for Disordered Energy Balance in Polycystic Ovary Syndrome
- PMID: 33383677
- PMCID: PMC7796281
- DOI: 10.3390/ijms22010243
Androgen Reduces Mitochondrial Respiration in Mouse Brown Adipocytes: A Model for Disordered Energy Balance in Polycystic Ovary Syndrome
Abstract
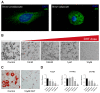
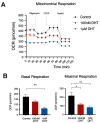
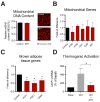
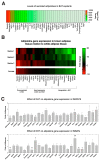
Polycystic ovary syndrome (PCOS) is a common endocrinopathy that is associated with an adverse metabolic profile including reduced postprandial thermogenesis. Although abnormalities in adipose tissue function have been widely reported in women with PCOS, less is known about direct effects of androgen on white and, particularly, brown adipocytes. The purpose of this study was to investigate the effect of the nonaromatizable androgen dihydrotestosterone (DHT) on (1) lipid accumulation and expression of adipogenic markers in immortalized mouse brown adipose cell lines (IMBATs), (2) mitochondrial respiration in IMBATs, (3) mitochondrial DNA content and gene expression, (4) expression of brown adipose tissue (BAT) markers and thermogenic activation. In addition, we profiled the relative levels of 38 adipokines secreted from BAT explants and looked at androgen effects on adipokine gene expression in both IMBATs and immortalized mouse white adipose (IMWATs) cell lines. Androgen treatment inhibited IMBAT differentiation in a dose-dependent manner, reduced markers of adipogenesis, and attenuated the β-adrenoceptor-stimulated increase in uncoupling protein-1 (UCP1) expression. In explants of mouse interscapular BAT, androgen reduced expression of UCP1, peroxisome proliferator-activated receptor-γ coactivator-1 (PCG-1) and Cidea. Significantly, as well as affecting genes involved in thermogenesis in BAT, androgen treatment reduced mitochondrial respiration in IMBATs, as measured by the Seahorse XF method. The results of this study suggest a role for excess androgen in inhibiting brown adipogenesis, attenuating the activation of thermogenesis and reducing mitochondrial respiration in BAT. Together, these data provide a plausible molecular mechanism that may contribute to reduced postprandial thermogenesis and the tendency to obesity in women with PCOS.
Keywords: PCOS; UCP1; androgens; brown adipose tissue; mitochondrial respiration.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
MicroRNA-21 modulates brown adipose tissue adipogenesis and thermogenesis in a mouse model of polycystic ovary syndrome.Biol Sex Differ. 2024 Jul 10;15(1):53. doi: 10.1186/s13293-024-00630-2. Biol Sex Differ. 2024. PMID: 38987854 Free PMC article.
-
Irisin exerts dual effects on browning and adipogenesis of human white adipocytes.Am J Physiol Endocrinol Metab. 2016 Aug 1;311(2):E530-41. doi: 10.1152/ajpendo.00094.2016. Epub 2016 Jul 19. Am J Physiol Endocrinol Metab. 2016. PMID: 27436609
-
Brown Adipose Tissue and Novel Management Strategies for Polycystic Ovary Syndrome Therapy.Front Endocrinol (Lausanne). 2022 May 19;13:847249. doi: 10.3389/fendo.2022.847249. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35663310 Free PMC article. Review.
-
miR-199a-3p regulates brown adipocyte differentiation through mTOR signaling pathway.Mol Cell Endocrinol. 2018 Nov 15;476:155-164. doi: 10.1016/j.mce.2018.05.005. Epub 2018 Jun 28. Mol Cell Endocrinol. 2018. PMID: 29753771
-
Origins and early development of the concept that brown adipose tissue thermogenesis is linked to energy balance and obesity.Biochimie. 2017 Mar;134:62-70. doi: 10.1016/j.biochi.2016.09.007. Epub 2016 Sep 10. Biochimie. 2017. PMID: 27621146 Review.
Cited by
-
Sex Differences in Brown Adipose Tissue Function: Sex Hormones, Glucocorticoids, and Their Crosstalk.Front Endocrinol (Lausanne). 2021 Apr 13;12:652444. doi: 10.3389/fendo.2021.652444. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 33927694 Free PMC article. Review.
-
The Implication of Mechanistic Approaches and the Role of the Microbiome in Polycystic Ovary Syndrome (PCOS): A Review.Metabolites. 2023 Jan 14;13(1):129. doi: 10.3390/metabo13010129. Metabolites. 2023. PMID: 36677054 Free PMC article. Review.
-
Circulating GDF15 concentrations in girls with low birth weight: effects of prolonged metformin treatment.Pediatr Res. 2023 Mar;93(4):964-968. doi: 10.1038/s41390-022-02175-9. Epub 2022 Jul 11. Pediatr Res. 2023. PMID: 35817957
-
Mechanistic insights into the Bushen Huatan Huoxue Formula and its components in ameliorating obesity-associated polycystic ovary syndrome.Chin Med. 2025 Jul 1;20(1):99. doi: 10.1186/s13020-025-01165-3. Chin Med. 2025. PMID: 40597295 Free PMC article.
-
Castration promotes the browning of the prostate tumor microenvironment.Cell Commun Signal. 2023 Sep 28;21(1):267. doi: 10.1186/s12964-023-01294-y. Cell Commun Signal. 2023. PMID: 37770940 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

