Synthesis of Galectin Inhibitors by Regioselective 3'- O-Sulfation of Vanillin Lactosides Obtained under Phase Transfer Catalysis
- PMID: 33383774
- PMCID: PMC7795656
- DOI: 10.3390/molecules26010115
Synthesis of Galectin Inhibitors by Regioselective 3'- O-Sulfation of Vanillin Lactosides Obtained under Phase Transfer Catalysis
Abstract
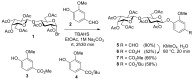
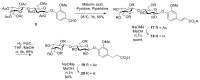
Vanillin-based lactoside derivatives were synthetized using phase-transfer catalyzed reactions from per-O-acetylated lactosyl bromide. The aldehyde group of the vanillin moiety was then modified to generate a series of related analogs having variable functionalities in the para- position of the aromatic residue. The corresponding unprotected lactosides, obtained by Zemplén transesterification, were regioselectively 3'-O-sulfated using tin chemistry activation followed by treatment with sulfur trioxide-trimethylamine complex (Men3N-SO3). Additional derivatives were also prepared from the vanillin's aldehyde using a Knoevenagel reaction to provide extended α, β-unsaturated carboxylic acid which was next reduced to the saturated counterpart.
Keywords: Knoevenagel-Doebner reaction; cancer; galectins; lactosides; phase transfer catalysis; sulfation; vanillin.
Conflict of interest statement
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results
Figures

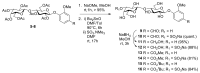
Similar articles
-
Defining the potential of aglycone modifications for affinity/selectivity enhancement against medically relevant lectins: synthesis, activity screening, and HSQC-based NMR analysis.Chembiochem. 2015 Jan 2;16(1):126-39. doi: 10.1002/cbic.201402474. Epub 2014 Nov 18. Chembiochem. 2015. PMID: 25407851
-
Synthesis of regioselectively sulfated xylodextrins and crystal structure of sodium methyl beta-D-xylopyranoside 4-O-sulfate hemihydrate.Carbohydr Res. 2009 Jan 5;344(1):21-8. doi: 10.1016/j.carres.2008.09.018. Epub 2008 Sep 24. Carbohydr Res. 2009. PMID: 18996512
-
Synthesis of stable and selective inhibitors of human galectins-1 and -3.Bioorg Med Chem. 2008 Aug 15;16(16):7811-23. doi: 10.1016/j.bmc.2008.06.044. Epub 2008 Jun 26. Bioorg Med Chem. 2008. PMID: 18674915
-
Synthesis of bivalent lactosides based on terephthalamide, N,N'-diglucosylterephthalamide, and glycophane scaffolds and assessment of their inhibitory capacity on medically relevant lectins.J Org Chem. 2009 Dec 4;74(23):9010-26. doi: 10.1021/jo901667r. J Org Chem. 2009. PMID: 19899775
-
Therapeutic Potential of Vanillin and its Main Metabolites to Regulate the Inflammatory Response and Oxidative Stress.Mini Rev Med Chem. 2019;19(20):1681-1693. doi: 10.2174/1389557519666190312164355. Mini Rev Med Chem. 2019. PMID: 30864521 Review.
Cited by
-
Selectively Modified Lactose and N-Acetyllactosamine Analogs at Three Key Positions to Afford Effective Galectin-3 Ligands.Int J Mol Sci. 2023 Feb 13;24(4):3718. doi: 10.3390/ijms24043718. Int J Mol Sci. 2023. PMID: 36835132 Free PMC article.
-
Recent Development in the Design of Neoglycoliposomes Bearing Arborescent Architectures.Molecules. 2021 Jul 15;26(14):4281. doi: 10.3390/molecules26144281. Molecules. 2021. PMID: 34299556 Free PMC article. Review.
References
-
- Solís D., Bovin N.V., Davis A.P., Jiménez-Barbero J., Romero A., Roy R., Smetana Jr K., Gabius H.-J. A guide into glycosciences: How chemistry and biochemistry cooperate to crack the sugar code. Biochim. Biophys. Acta. 2015;1850:186–235. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

