The Neurotransmitters Involved in Drosophila Alcohol-Induced Behaviors
- PMID: 33384590
- PMCID: PMC7770116
- DOI: 10.3389/fnbeh.2020.607700
The Neurotransmitters Involved in Drosophila Alcohol-Induced Behaviors
Abstract
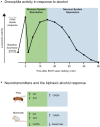
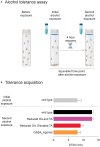
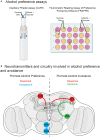
Alcohol is a widely used and abused substance with numerous negative consequences for human health and safety. Historically, alcohol's widespread, non-specific neurobiological effects have made it a challenge to study in humans. Therefore, model organisms are a critical tool for unraveling the mechanisms of alcohol action and subsequent effects on behavior. Drosophila melanogaster is genetically tractable and displays a vast behavioral repertoire, making it a particularly good candidate for examining the neurobiology of alcohol responses. In addition to being experimentally amenable, Drosophila have high face and mechanistic validity: their alcohol-related behaviors are remarkably consistent with humans and other mammalian species, and they share numerous conserved neurotransmitters and signaling pathways. Flies have a long history in alcohol research, which has been enhanced in recent years by the development of tools that allow for manipulating individual Drosophila neurotransmitters. Through advancements such as the GAL4/UAS system and CRISPR/Cas9 mutagenesis, investigation of specific neurotransmitters in small subsets of neurons has become ever more achievable. In this review, we describe recent progress in understanding the contribution of seven neurotransmitters to fly behavior, focusing on their roles in alcohol response: dopamine, octopamine, tyramine, serotonin, glutamate, GABA, and acetylcholine. We chose these small-molecule neurotransmitters due to their conservation in mammals and their importance for behavior. While neurotransmitters like dopamine and octopamine have received significant research emphasis regarding their contributions to behavior, others, like glutamate, GABA, and acetylcholine, remain relatively unexplored. Here, we summarize recent genetic and behavioral findings concerning these seven neurotransmitters and their roles in the behavioral response to alcohol, highlighting the fitness of the fly as a model for human alcohol use.
Keywords: AUD; Drosophila; alcohol abuse; alcohol behavior; genetics; neurotransmitter.
Copyright © 2020 Chvilicek, Titos and Rothenfluh.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

