Biochemical Timekeeping Via Reentrant Phase Transitions
- PMID: 33387533
- PMCID: PMC8154630
- DOI: 10.1016/j.jmb.2020.166794
Biochemical Timekeeping Via Reentrant Phase Transitions
Abstract
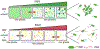
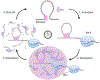
Appreciation for the role of liquid-liquid phase separation in the functional organization of cellular matter has exploded in recent years. More recently there has been a growing effort to understand the principles of heterotypic phase separation, the demixing of multiple proteins and nucleic acids into a single functional condensate. A phase transition is termed reentrant if it involves the transformation of a system from one state into a macroscopically similar or identical state via at least two phase transitions elicited by variation of a single parameter. Reentrant liquid-liquid phase separation can occur when the condensation of one species is tuned by another. Reentrant phase transitions have been modeled in vitro using protein and RNA mixtures. These biochemical studies reveal two features of reentrant phase separation that are likely important to functional cellular condensates: (1) the ability to generate condensates with layered functional topologies, and (2) the ability to generate condensates whose composition and duration are self-limiting to enable a form of biochemical timekeeping. We relate these biochemical studies to potential cellular examples and discuss how layered topologies and self-regulation may impact key biological processes.
Keywords: RNA; condensates; disordered proteins; phase separation; reentrant phase transitions; transcription.
Copyright © 2020 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest J.S. is a consultant for Dewpoint Therapeutics and Maze Therapeutics.
Figures
Similar articles
-
Phase Separation in Mixtures of Prion-Like Low Complexity Domains is Driven by the Interplay of Homotypic and Heterotypic Interactions.bioRxiv [Preprint]. 2023 Mar 16:2023.03.15.532828. doi: 10.1101/2023.03.15.532828. bioRxiv. 2023. Update in: Nat Commun. 2023 Sep 8;14(1):5527. doi: 10.1038/s41467-023-41274-x. PMID: 36993212 Free PMC article. Updated. Preprint.
-
Interplay between Short-Range Attraction and Long-Range Repulsion Controls Reentrant Liquid Condensation of Ribonucleoprotein-RNA Complexes.J Am Chem Soc. 2019 Sep 18;141(37):14593-14602. doi: 10.1021/jacs.9b03689. Epub 2019 Sep 5. J Am Chem Soc. 2019. PMID: 31437398 Free PMC article.
-
Using quantitative reconstitution to investigate multicomponent condensates.RNA. 2022 Jan;28(1):27-35. doi: 10.1261/rna.079008.121. Epub 2021 Nov 12. RNA. 2022. PMID: 34772789 Free PMC article. Review.
-
Temperature-dependent reentrant phase transition of RNA-polycation mixtures.Soft Matter. 2022 Feb 16;18(7):1342-1349. doi: 10.1039/d1sm01557e. Soft Matter. 2022. PMID: 34984429 Free PMC article.
-
Analysis of biomolecular condensates and protein phase separation with microfluidic technology.Biochim Biophys Acta Mol Cell Res. 2021 Jan;1868(1):118823. doi: 10.1016/j.bbamcr.2020.118823. Epub 2020 Aug 13. Biochim Biophys Acta Mol Cell Res. 2021. PMID: 32800925 Review.
Cited by
-
Targeting nucleic acid phase transitions as a mechanism of action for antimicrobial peptides.Nat Commun. 2023 Nov 7;14(1):7170. doi: 10.1038/s41467-023-42374-4. Nat Commun. 2023. PMID: 37935659 Free PMC article.
-
Reentrant condensation of a multicomponent cola/milk system induced by polyphosphate.Food Chem X. 2024 Jan 28;21:101165. doi: 10.1016/j.fochx.2024.101165. eCollection 2024 Mar 30. Food Chem X. 2024. PMID: 38328695 Free PMC article.
-
Merging Established Mechanisms with New Insights: Condensates, Hubs, and the Regulation of RNA Polymerase II Transcription.J Mol Biol. 2022 Jan 15;434(1):167216. doi: 10.1016/j.jmb.2021.167216. Epub 2021 Aug 30. J Mol Biol. 2022. PMID: 34474085 Free PMC article. Review.
-
Assembly of the Tripartite and RNA Condensates of the Respiratory Syncytial Virus Factory Proteins In Vitro: Role of the Transcription Antiterminator M2-1.Viruses. 2023 Jun 6;15(6):1329. doi: 10.3390/v15061329. Viruses. 2023. PMID: 37376628 Free PMC article.
-
Melatonin: Regulation of Prion Protein Phase Separation in Cancer Multidrug Resistance.Molecules. 2022 Jan 21;27(3):705. doi: 10.3390/molecules27030705. Molecules. 2022. PMID: 35163973 Free PMC article. Review.
References
-
- Patel A, Lee HO, Jawerth L, Maharana S, Jahnel M, Hein MY, Stoynov S, Mahamid J, Saha S, Franzmann TM, Pozniakovski A, Poser I, Maghelli N, Royer LA, Weigert M, Myers EW, Grill S, Drechsel D, Hyman AA, Alberti S, A Liquid-to-Solid Phase Transition of the ALS Protein FUS Accelerated by Disease Mutation, Cell 162 (2015) 1066–1077. - PubMed
-
- Mann JR, Gleixner AM, Mauna JC, Gomes E, DeChellis-Marks MR, Needham PG, Copley KE, Hurtle B, Portz B, Pyles NJ, Guo L, Calder CB, Wills ZP, Pandey UB, Kofler JK, Brodsky JL, Thathiah A, Shorter J, Donnelly CJ, RNA Binding Antagonizes Neurotoxic Phase Transitions of TDP-43, Neuron 102 (2019) 321–338.e8. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

