Screening for the prevention and early detection of cervical cancer: protocol for systematic reviews to inform Canadian recommendations
- PMID: 33388083
- PMCID: PMC7777363
- DOI: 10.1186/s13643-020-01538-9
Screening for the prevention and early detection of cervical cancer: protocol for systematic reviews to inform Canadian recommendations
Abstract
Purpose: To inform recommendations by the Canadian Task Force on Preventive Health Care on screening in primary care for the prevention and early detection of cervical cancer by systematically reviewing evidence of (a) effectiveness; (b) test accuracy; (c) individuals' values and preferences; and (d) strategies aimed at improving screening rates.
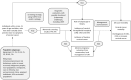
Methods: De novo reviews will be conducted to evaluate effectiveness and to assess values and preferences. For test accuracy and strategies to improve screening rates, we will integrate studies from existing systematic reviews with search updates to the present. Two Cochrane reviews will provide evidence of adverse pregnancy outcomes from the conservative management of cervical intraepithelial neoplasia. We will search Medline, Embase, and Cochrane Central (except for individuals' values and preferences, where Medline, Scopus, and EconLit will be searched) via peer-reviewed search strategies and the reference lists of included studies and reviews. We will search ClinicalTrials.gov and the World Health Organization International Clinical Trials Registry Platform for ongoing trials. Two reviewers will screen potentially eligible studies and agree on those to include. Data will be extracted by one reviewer with verification by another. Two reviewers will independently assess risk of bias and reach consensus. Where possible and suitable, we will pool studies via meta-analysis. We will compare accuracy data per outcome and per comparison using the Rutter and Gatsonis hierarchical summary receiver operating characteristic model and report relative sensitivities and specificities. Findings on values and preferences will be synthesized using a narrative synthesis approach and thematic analysis, depending on study designs. Two reviewers will appraise the certainty of evidence for all outcomes using GRADE (Grading of Recommendations Assessment, Development and Evaluation) and come to consensus.
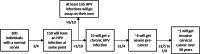
Discussion: The publication of guidance on screening in primary care for the prevention and early detection of cervical cancer by the Task Force in 2013 focused on cytology. Since 2013, new studies using human papillomavirus tests for cervical screening have been published that will improve our understanding of screening in primary care settings. This review will inform updated recommendations based on currently available studies and address key evidence gaps noted in our previous review.
Keywords: Cervical intraepithelial neoplasia; Guideline; Mass screening; Primary health care; Systematic review; Uterine cervical neoplasms.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
-
- Canadian Cancer Society . Cervical cancer. 2019.
-
- Herrero R. Cervical cancer. In: Thun MJ, Linet MS, Cerhan JR, Haiman CA, Schottenfeld D, editors. Cancer epidemiology and prevention. 4. New York: Oxford University Press; 2018. pp. 925–946.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical