Locomotor deficits induced by lumbar muscle inflammation involve spinal microglia and are independent of KCC2 expression in a mouse model of complete spinal transection
- PMID: 33388315
- PMCID: PMC7904639
- DOI: 10.1016/j.expneurol.2020.113592
Locomotor deficits induced by lumbar muscle inflammation involve spinal microglia and are independent of KCC2 expression in a mouse model of complete spinal transection
Abstract
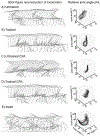
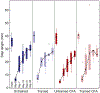
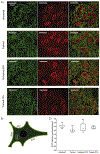
Spinal cord injury (SCI) is associated with damage to musculoskeletal tissues of the spine. Recent findings show that pain and inflammatory processes caused by musculoskeletal injury mediate plastic changes in the spinal cord. These changes could impede the adaptive plastic changes responsible for functional recovery. The underlying mechanism remains unclear, but may involve the microglia-BDNF-KCC2 pathway, which is implicated in sensitization of dorsal horn neurons in neuropathic pain and in the regulation of spinal excitability by step-training. In the present study, we examined the effects of step-training and lumbar muscle inflammation induced by complete Freund's adjuvant (CFA) on treadmill locomotion in a mouse model of complete spinal transection. The impact on locomotor recovery of each of these interventions alone or in combination were examined in addition to changes in microglia and KCC2 expression in the dorsal and ventral horns of the sublesional spinal cord. Results show that angular motion at the hip, knee and ankle joint during locomotion were decreased by CFA injection and improved by step-training. Moreover, CFA injection enhanced the expression of the microglial marker Iba1 in both ventral and dorsal horns, with or without step-training. However, this change was not associated with a modulation of KCC2 expression, suggesting that locomotor deficits induced by inflammation are independent of KCC2 expression in the sublesional spinal cord. These results indicate that musculoskeletal injury hinders locomotor recovery after SCI and that microglia is involved in this effect.
Keywords: Chloride homeostasis; Disinhibition; Neuroplasticity; Nociception; Pain; Spinal cord injury model; Training.
Crown Copyright © 2021. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
AUTHORS DISCLOSE STATEMENT
No competing financial interests exist.
Figures



Similar articles
-
Lumbar muscle inflammation alters spinally mediated locomotor recovery induced by training in a mouse model of complete spinal cord injury.Neuroscience. 2017 Sep 17;359:69-81. doi: 10.1016/j.neuroscience.2017.07.010. Epub 2017 Jul 15. Neuroscience. 2017. PMID: 28716590
-
H-reflex disinhibition by lumbar muscle inflammation in a mouse model of spinal cord injury.Neurosci Lett. 2019 Jan 18;690:36-41. doi: 10.1016/j.neulet.2018.10.005. Epub 2018 Oct 4. Neurosci Lett. 2019. PMID: 30292718
-
Epigenetic suppression of potassium-chloride co-transporter 2 expression in inflammatory pain induced by complete Freund's adjuvant (CFA).Eur J Pain. 2017 Feb;21(2):309-321. doi: 10.1002/ejp.925. Epub 2016 Aug 10. Eur J Pain. 2017. PMID: 27506893
-
Learning to promote recovery after spinal cord injury.Exp Neurol. 2020 Aug;330:113334. doi: 10.1016/j.expneurol.2020.113334. Epub 2020 Apr 28. Exp Neurol. 2020. PMID: 32353465 Free PMC article. Review.
-
The effects and potential mechanisms of locomotor training on improvements of functional recovery after spinal cord injury.Int Rev Neurobiol. 2019;147:199-217. doi: 10.1016/bs.irn.2019.08.003. Epub 2019 Sep 16. Int Rev Neurobiol. 2019. PMID: 31607355 Review.
Cited by
-
Role of Exercise on Inflammation Cytokines of Neuropathic Pain in Animal Models.Mol Neurobiol. 2024 Dec;61(12):10288-10301. doi: 10.1007/s12035-024-04214-4. Epub 2024 May 7. Mol Neurobiol. 2024. PMID: 38714582 Review.
-
Immunoregulation of Glia after spinal cord injury: a bibliometric analysis.Front Immunol. 2024 Jun 13;15:1402349. doi: 10.3389/fimmu.2024.1402349. eCollection 2024. Front Immunol. 2024. PMID: 38938572 Free PMC article.
-
Temporal changes of spinal microglia in murine models of neuropathic pain: a scoping review.Front Immunol. 2024 Dec 6;15:1460072. doi: 10.3389/fimmu.2024.1460072. eCollection 2024. Front Immunol. 2024. PMID: 39735541 Free PMC article.
-
Tuina alleviates neuropathic pain through regulate the activation of microglia and the secretion of inflammatory cytokine in spinal cord.J Tradit Chin Med. 2024 Aug;44(4):762-769. doi: 10.19852/j.cnki.jtcm.20240515.002. J Tradit Chin Med. 2024. PMID: 39066537 Free PMC article.
References
-
- Barrière G, Leblond H, Provencher J, Rossignol S, 2008. Prominent Role of the Spinal Central Pattern Generator in the Recovery of Locomotion after Partial Spinal Cord Injuries. Journal of Neuroscience 28, 3976–3987. https://10.1523/jneurosci.5692-07.2008. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

