Implementation of a screening tool for primary ciliary dyskinesia (PCD) in a pediatric otolaryngology clinic
- PMID: 33388601
- PMCID: PMC8021076
- DOI: 10.1016/j.ijporl.2020.110586
Implementation of a screening tool for primary ciliary dyskinesia (PCD) in a pediatric otolaryngology clinic
Abstract
Background: Primary ciliary dyskinesia (PCD) is a rare genetic disease arising from motile ciliary dysfunction and associated with recurrent and chronic upper and lower respiratory tract infections. Pediatric otolaryngologists may see these patients prior to the development of lung disease. Features of PCD may overlap with other suppurative respiratory diseases, creating diagnostic challenges. A simple screening tool would be beneficial to identify potential patients who have chronic upper respiratory tract disease requiring further specialist evaluation.
Objective: To test a simple screening tool consisting of four questions to detect PCD in children with chronic otitis media and chronic rhinosinusitis seen in a tertiary otolaryngology clinic.
Methods: A prospective, single site, observational study in a tertiary care pediatric otolaryngology clinic. Children aged 3-17 years diagnosed with chronic otitis media or rhinosinusitis with onset at less than 2 years of age were recruited. All study subjects had at least one of four key clinical features for PCD as determined by answers to screening questions, while control subjects had none. All participants completed a medical history questionnaire and nasal nitric oxide measurements. Those with reduced nasal nitric oxide levels were referred to our PCD center for further evaluation.
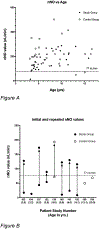
Results: A total of 153 patients were screened and 62 subjects were enrolled. Of those, 35 were enrolled as study subjects and 27 as matched controls. Study subjects had mean age of 7.5 years (3.2-16.5) with pre-screening diagnosis of chronic otitis media (n = 29) or chronic rhinosinusitis (n = 6). Control subjects (n = 27) had mean age 7.2 years (3.0-16.3) with pre-screening diagnosis of chronic otitis media (n = 25), and chronic rhinosinusitis (n = 2). There were no differences in subject demographics or mean nasal nitric oxide values between the two groups (179.8 vs 210.8 nl/min). Ten individuals had low nasal nitric oxide values, 7 of which were normal on repeat testing. Three subjects failed to return for follow up evaluations. Four referrals were made for further evaluation on the basis of clinical symptoms and nasal nitric oxide results. While no new cases of PCD were detected, a subject and his sibling with recurrent sinopulmonary infections were referred for immunologic evaluation.
Conclusion: The use of standardized screening questions can be used in an otolaryngology clinic to identify patients who require further evaluation for PCD or primary immunodeficiency.
Keywords: Children; Early diagnosis; Nasal nitric oxide; Primary ciliary dyskinesia; Primary immunodeficiency.
Copyright © 2020 Elsevier B.V. All rights reserved.
Figures
Similar articles
-
Nasal Nitric Oxide in Primary Immunodeficiency and Primary Ciliary Dyskinesia: Helping to Distinguish Between Clinically Similar Diseases.J Clin Immunol. 2019 Feb;39(2):216-224. doi: 10.1007/s10875-019-00613-8. Epub 2019 Mar 26. J Clin Immunol. 2019. PMID: 30911954 Free PMC article.
-
Nasal nitric oxide levels in primary ciliary dyskinesia, cystic fibrosis and healthy children.Turk J Pediatr. 2019;61(1):20-25. doi: 10.24953/turkjped.2019.01.004. Turk J Pediatr. 2019. PMID: 31559717 Clinical Trial.
-
Clinical Features and Associated Likelihood of Primary Ciliary Dyskinesia in Children and Adolescents.Ann Am Thorac Soc. 2016 Aug;13(8):1305-13. doi: 10.1513/AnnalsATS.201511-748OC. Ann Am Thorac Soc. 2016. PMID: 27070726 Free PMC article.
-
Recent advances in primary ciliary dyskinesia.Auris Nasus Larynx. 2016 Jun;43(3):229-36. doi: 10.1016/j.anl.2015.09.012. Epub 2015 Oct 31. Auris Nasus Larynx. 2016. PMID: 26527516 Review.
-
[Primary ciliary dyskinesia: a retrospective review of clinical and paraclinical data].Rev Mal Respir. 2011 Sep;28(7):856-63. doi: 10.1016/j.rmr.2011.02.014. Epub 2011 Sep 3. Rev Mal Respir. 2011. PMID: 21943530 Review. French.
Cited by
-
AggreBots: configuring CiliaBots through guided, modular tissue aggregation.bioRxiv [Preprint]. 2025 Feb 27:2025.02.22.639695. doi: 10.1101/2025.02.22.639695. bioRxiv. 2025. PMID: 40060463 Free PMC article. Preprint.
-
Engineering rotating apical-out airway organoid for assessing respiratory cilia motility.iScience. 2022 Jul 9;25(8):104730. doi: 10.1016/j.isci.2022.104730. eCollection 2022 Aug 19. iScience. 2022. PMID: 35942088 Free PMC article.
References
-
- Loges NT, Antony D, Maver A, Deardorff MA, Güleç EY, Gezdirici A, Nöthe-Menchen T, Höben IM, Jelten L, Frank D, Werner C, Tebbe J, Wu K, Goldmuntz E, Čuturilo G, Krock B, Ritter A, Hjeij R, Bakey Z, Pennekamp P, Dworniczak B, Brunner H, Peterlin B, Tanidir C, Olbrich H, Omran H, Schmidts M, Recessive DNAH9 loss-of-function mutations cause laterality defects and subtle respiratory ciliary-beating defects, Am. J. Hum. Genet (2018). 10.1016/j.ajhg.2018.10.020. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

