Primary Cilia, Ciliogenesis and the Actin Cytoskeleton: A Little Less Resorption, A Little More Actin Please
- PMID: 33392209
- PMCID: PMC7773788
- DOI: 10.3389/fcell.2020.622822
Primary Cilia, Ciliogenesis and the Actin Cytoskeleton: A Little Less Resorption, A Little More Actin Please
Abstract
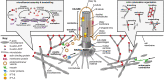
Primary cilia are microtubule-based organelles that extend from the apical surface of most mammalian cells, forming when the basal body (derived from the mother centriole) docks at the apical cell membrane. They act as universal cellular "antennae" in vertebrates that receive and integrate mechanical and chemical signals from the extracellular environment, serving diverse roles in chemo-, mechano- and photo-sensation that control developmental signaling, cell polarity and cell proliferation. Mutations in ciliary genes cause a major group of inherited developmental disorders called ciliopathies. There are very few preventative treatments or new therapeutic interventions that modify disease progression or the long-term outlook of patients with these conditions. Recent work has identified at least four distinct but interrelated cellular processes that regulate cilia formation and maintenance, comprising the cell cycle, cellular proteostasis, signaling pathways and structural influences of the actin cytoskeleton. The actin cytoskeleton is composed of microfilaments that are formed from filamentous (F) polymers of globular G-actin subunits. Actin filaments are organized into bundles and networks, and are attached to the cell membrane, by diverse cross-linking proteins. During cell migration, actin filament bundles form either radially at the leading edge or as axial stress fibers. Early studies demonstrated that loss-of-function mutations in ciliopathy genes increased stress fiber formation and impaired ciliogenesis whereas pharmacological inhibition of actin polymerization promoted ciliogenesis. These studies suggest that polymerization of the actin cytoskeleton, F-actin branching and the formation of stress fibers all inhibit primary cilium formation, whereas depolymerization or depletion of actin enhance ciliogenesis. Here, we review the mechanistic basis for these effects on ciliogenesis, which comprise several cellular processes acting in concert at different timescales. Actin polymerization is both a physical barrier to both cilia-targeted vesicle transport and to the membrane remodeling required for ciliogenesis. In contrast, actin may cause cilia loss by localizing disassembly factors at the ciliary base, and F-actin branching may itself activate the YAP/TAZ pathway to promote cilia disassembly. The fundamental role of actin polymerization in the control of ciliogenesis may present potential new targets for disease-modifying therapeutic approaches in treating ciliopathies.
Keywords: ROCK inhibitors; actin cytoskeleton; cilia; ciliogenesis; ciliopathies; cytoskeletal drugs; drug screen; polycystic kidney disease.
Copyright © 2020 Smith, Lake and Johnson.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Ajzeberg H., Slaats G., Stokman M., Logister I., Knoers N., Giles R. (2015). Urine-derived Renal Epithelial Cells (URECs) as a source of biomaterial from ciliopathy patients for functional studies and diagnostics. Cilia 4 51–51. 10.1186/2046-2530-4-S1-P51 - DOI
Publication types
LinkOut - more resources
Full Text Sources

