Neurotransmitter Release Site Replenishment and Presynaptic Plasticity
- PMID: 33396919
- PMCID: PMC7794938
- DOI: 10.3390/ijms22010327
Neurotransmitter Release Site Replenishment and Presynaptic Plasticity
Abstract
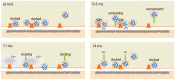
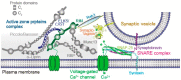
An action potential (AP) triggers neurotransmitter release from synaptic vesicles (SVs) docking to a specialized release site of presynaptic plasma membrane, the active zone (AZ). The AP simultaneously controls the release site replenishment with SV for sustainable synaptic transmission in response to incoming neuronal signals. Although many studies have suggested that the replenishment time is relatively slow, recent studies exploring high speed resolution have revealed SV dynamics with milliseconds timescale after an AP. Accurate regulation is conferred by proteins sensing Ca2+ entering through voltage-gated Ca2+ channels opened by an AP. This review summarizes how millisecond Ca2+ dynamics activate multiple protein cascades for control of the release site replenishment with release-ready SVs that underlie presynaptic short-term plasticity.
Keywords: Ca2+ channels; Ca2+ sensors; action potential; active zone; dynamin; myosin; presynaptic plasticity; presynaptic proteins; synaptic vesicle.
Conflict of interest statement
The author declares no conflict of interest.
Figures





References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

