Exploring healthcare professionals' knowledge, attitude, and practices towards pharmacovigilance: a cross-sectional survey
- PMID: 33397478
- PMCID: PMC7784002
- DOI: 10.1186/s40545-020-00287-3
Exploring healthcare professionals' knowledge, attitude, and practices towards pharmacovigilance: a cross-sectional survey
Abstract
Background: Spontaneous reporting of adverse drug reactions (ADRs) is a method of monitoring the safety of drugs and is the basic strategy for the post-marketing surveillance of the suspected drugs. Despite its importance, there is very little reporting of ADRs by healthcare professionals. The present study has evaluated the knowledge, attitude and practices of health care professionals (HCPs) regarding pharmacovigilance activities in Lahore, Pakistan.
Methods: A cross-sectional questionnaire-based survey was employed, and a convenience sampling was opted to collect the data among physicians, pharmacists and nurses working in tertiary care public hospitals of Lahore, Pakistan from September 2018 to January 2019.
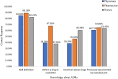
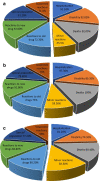
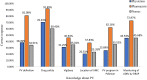
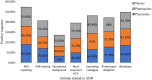
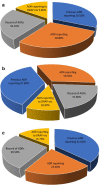
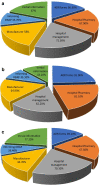
Results: Of the 384 questionnaires distributed, 346 health care professionals responded to the questionnaire (90.10% response rate). Most participants had good knowledge about ADR reporting, but pharmacist had comparatively better knowledge than other HCPs regarding ADR (89.18%) pharmacovigilance system (81.08%), its centres (72.97%) and function (91.89%). Most of the participants exhibited positive attitude regarding ADR reporting, such as 49.1% of physicians (P < 0.05), 70.2% pharmacists and 76.1% nurses showed a positive attitude that they are the most important HCPs to report an ADR. About 64.3% of physicians (P < 0.05) emphasized that consulting other colleagues is important before reporting an ADR. Of all, 77.7% physicians, 75.7% pharmacists and 68% of nurses had positive attitude that ADR reporting is a professional obligation and 67.6% of the pharmacists stated that they have reported ADRs in their workplace and 77.2% nurses have verbally reported ADRs to the concerned personnel or department.
Conclusion: Among all HCPs, pharmacists had better knowledge about ADR reporting and pharmacovigilance. All HCPs had positive attitude and inclination towards ADR reporting. The discrepancies were observed in the practices related to ADR reporting, whereas most of the participants including physicians and nurses did not report any ADR. Based on the above, strategies are needed to educate, train, and empower the HCPs in the domain of pharmacovigilance.
Keywords: Adverse drug reaction; Adverse drug reaction reporting; Drug regulatory authority of Pakistan; Healthcare professionals; Medicines safety; Pharmacovigilance.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
-
- Hussain R, Hassali MA, Babar ZUD. Medicines safety in the globalized context. Global pharmaceutical policy. Singapore: Springer; 2020. pp. 1–28.
-
- Wiholm BE, Olsson S, Moore N, Waller P. Spontaneous reporting systems outside the US. In: Strom BL, editor. Pharmacoepidemiology. 3. Chichester: Wiley; 2000. pp. 175–92.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
