Differential Contribution of Cadm1-Cadm3 Cell Adhesion Molecules to Peripheral Myelinated Axons
- PMID: 33397712
- PMCID: PMC7896006
- DOI: 10.1523/JNEUROSCI.2736-20.2020
Differential Contribution of Cadm1-Cadm3 Cell Adhesion Molecules to Peripheral Myelinated Axons
Abstract
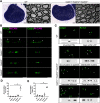
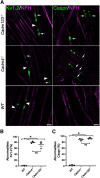
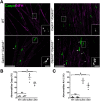
Cell adhesion proteins of the Cadm (SynCAM/Necl) family regulate myelination and the organization of myelinated axons. In the peripheral nervous system (PNS), intercellular contact between Schwann cells and their underlying axons is believed to be mediated by binding of glial Cadm4 to axonal Cadm3 or Cadm2. Nevertheless, given that distinct neurons express different combinations of the Cadm proteins, the identity of the functional axonal ligand for Cadm4 remains to be determined. Here, we took a genetic approach to compare the phenotype of Cadm4 null mice, which exhibit abnormal distribution of Caspr and Kv1 potassium channels, with mice lacking different combinations of Cadm1-Cadm3 genes. We show that in contrast to mice lacking the single Cadm1, Cadm2, or Cadm3 genes, genetic ablation of all three phenocopies the abnormalities detected in the absence of Cadm4. Similar defects were observed in double mutant mice lacking Cadm3 and Cadm2 (i.e., Cadm3-/-/Cadm2-/-) or Cadm3 and Cadm1 (i.e., Cadm3-/-/Cadm1-/-), but not in mice lacking Cadm1 and Cadm2 (i.e., Cadm1-/-/Cadm2-/-). Furthermore, axonal organization abnormalities were also detected in Cadm3 null mice that were heterozygous for the two other axonal Cadms. Our results identify Cadm3 as the main axonal ligand for glial Cadm4, and reveal that its absence could be compensated by the combined action of Cadm2 and Cadm1.SIGNIFICANCE STATEMENT Myelination by Schwann cells enables fast conduction of action potentials along motor and sensory axons. In these nerves, Schwann cell-axon contact is mediated by cell adhesion molecules of the Cadm family. Cadm4 in Schwann cells regulates axonal ensheathment and myelin wrapping, as well as the organization of the axonal membrane, but the identity of its axonal ligands is not clear. Here, we reveal that Cadm mediated axon-glia interactions depend on a hierarchical adhesion code that involves multiple family members. Our results provide important insights into the molecular mechanisms of axon-glia communication, and the function of Cadm proteins in PNS myelin.
Keywords: Schwann cells; SynCAM; axon-glia interaction; cell adhesion; myelin; node of Ranvier.
Copyright © 2021 the authors.
Figures


Similar articles
-
Genetic deletion of Cadm4 results in myelin abnormalities resembling Charcot-Marie-Tooth neuropathy.J Neurosci. 2013 Jul 3;33(27):10950-61. doi: 10.1523/JNEUROSCI.0571-13.2013. J Neurosci. 2013. PMID: 23825401 Free PMC article.
-
A CADM3 variant causes Charcot-Marie-Tooth disease with marked upper limb involvement.Brain. 2021 May 7;144(4):1197-1213. doi: 10.1093/brain/awab019. Brain. 2021. PMID: 33889941 Free PMC article.
-
Cadm3 (Necl-1) interferes with the activation of the PI3 kinase/Akt signaling cascade and inhibits Schwann cell myelination in vitro.Glia. 2016 Dec;64(12):2247-2262. doi: 10.1002/glia.23072. Epub 2016 Sep 23. Glia. 2016. PMID: 27658374 Free PMC article.
-
Organization and maintenance of molecular domains in myelinated axons.J Neurosci Res. 2013 May;91(5):603-22. doi: 10.1002/jnr.23197. Epub 2013 Feb 13. J Neurosci Res. 2013. PMID: 23404451 Free PMC article. Review.
-
Axon-glial signaling and the glial support of axon function.Annu Rev Neurosci. 2008;31:535-61. doi: 10.1146/annurev.neuro.30.051606.094309. Annu Rev Neurosci. 2008. PMID: 18558866 Review.
Cited by
-
Restoration of spinal cord injury: From endogenous repairing process to cellular therapy.Front Cell Neurosci. 2022 Nov 29;16:1077441. doi: 10.3389/fncel.2022.1077441. eCollection 2022. Front Cell Neurosci. 2022. PMID: 36523818 Free PMC article. Review.
-
The CNV map construction and ROH analysis of Pinan cattle.BMC Genomics. 2025 May 14;26(1):480. doi: 10.1186/s12864-025-11626-6. BMC Genomics. 2025. PMID: 40369451 Free PMC article.
-
Single-Cell Transcriptomics and Lineage Tracing Unveil Parallels in Lymphatic Muscle and Venous Smooth Muscle Development, Identity, and Function.Arterioscler Thromb Vasc Biol. 2025 Jul;45(7):1207-1225. doi: 10.1161/ATVBAHA.125.322567. Epub 2025 May 15. Arterioscler Thromb Vasc Biol. 2025. PMID: 40371470 Free PMC article.
-
SynCAMs in Normal Vertebrate Neural Development and Neuropsychiatric Disorders: from the Perspective of the OCAs.Mol Neurobiol. 2024 Jan;61(1):358-371. doi: 10.1007/s12035-023-03579-2. Epub 2023 Aug 22. Mol Neurobiol. 2024. PMID: 37607992 Review.
-
Axonal Selectivity of Myelination by Single Oligodendrocytes Established During Development in Mouse Cerebellar White Matter.Glia. 2025 Apr;73(4):873-886. doi: 10.1002/glia.24660. Epub 2024 Dec 17. Glia. 2025. PMID: 39688302 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
