The protocol of a prospective, multicenter, randomized, controlled phase III study evaluating different cycles of oxaliplatin combined with S-1 (SOX) as neoadjuvant chemotherapy for patients with locally advanced gastric cancer: RESONANCE-II trial
- PMID: 33402102
- PMCID: PMC7786891
- DOI: 10.1186/s12885-020-07764-7
The protocol of a prospective, multicenter, randomized, controlled phase III study evaluating different cycles of oxaliplatin combined with S-1 (SOX) as neoadjuvant chemotherapy for patients with locally advanced gastric cancer: RESONANCE-II trial
Abstract
Background: Curing locally advanced gastric cancer through surgery alone is difficult. Adjuvant and neoadjuvant chemotherapy bring potential benefits to more patients with gastric cancer based on several clinical trials. According to phase II studies and guidelines, SOX regimen as neoadjuvant chemotherapy is efficient. However, the optimal duration of neoadjuvant chemotherapy has not been established. In this study, we will evaluate the efficacy and safety of different cycles of SOX as neoadjuvant chemotherapy for patients with locally advanced gastric cancer.
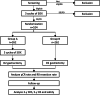
Methods: RESONANCE-II trial is a prospective, multicenter, randomized, controlled phase III study which will enroll 524 patients in total. Eligible patients will be registered, pre-enrolled and receive three cycles of SOX, after which tumor response evaluations will be carried out. Those who show stable disease or progressive disease will be excluded. Patients showing complete response or partial response will be enrolled and assigned into either group A for another three cycles of SOX (six cycles in total) followed by D2 surgery; or group B for D2 surgery (three cycles in total). The primary endpoint is the rate of pathological complete response and the secondary endpoints are R0 resection rate, three-year disease-free survival, five-year overall survival, and safety.
Discussion: This study is the first phase III randomized trial to compare the cycles of neoadjuvant chemotherapy using SOX for resectable locally advanced cancer. Based on a total of six to eight cycles of perioperative chemotherapy usually applied in locally advanced gastric cancer, patients in group A can be considered to have completed all perioperative chemotherapy, the results of which may suggest the feasibility of using chemotherapy only before surgery in gastric cancer.
Trial registration: Registered prospectively in the World Health Organization International Clinical Trials Registry Platform (WHO ICTRP) with registration number ChiCTR1900023293 on May 21st, 2019.
Keywords: Duration of neoadjuvant chemotherapy; Locally advanced gastric cancer; Oxaliplatin; S-1.
Conflict of interest statement
The authors declare that they have no competing interests.
References
-
- Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous