Modulation of heroin intake by ovarian hormones in gonadectomized and intact female rats
- PMID: 33404736
- PMCID: PMC8218341
- DOI: 10.1007/s00213-020-05743-1
Modulation of heroin intake by ovarian hormones in gonadectomized and intact female rats
Abstract
Rationale: Heroin intake decreases during the proestrus phase of the estrous cycle in female rats. Circulating concentrations of both estradiol and progesterone peak during proestrus, and it is not known which of these hormones, or their combination, are responsible for these effects.
Objectives: The purpose of this study was to determine the effects of estradiol, progesterone, and their combination on heroin self-administration in female rats.
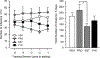
Methods: In Experiment 1, the estrous cycle of intact female rats was tracked daily. If a rat was in proestrus, either the estrogen receptor antagonist, raloxifene, the progesterone receptor antagonist, mifepristone, or their combination was administered 30 min prior to a heroin self-administration session. In Experiment 2, separate groups of ovariectomized female rats were treated chronically with exogenous estradiol, progesterone, estradiol + progesterone, or vehicle, and heroin intake was examined over a 100-fold dose range.
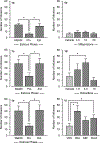
Results: In Experiment 1, raloxifene, but not mifepristone, significantly blocked proestrus-associated decreases in heroin intake. In Experiment 2, estrogentreated rats self-administered less heroin than any other group and significantly less heroin than rats treated with progesterone.
Conclusions: These data suggest that (1) estradiol but not progesterone is responsible for proestrus-associated decreases in heroin intake and (2) estradiol decreases heroin intake relative to progesterone. These data differ from those reported previously with stimulants and suggest that estrogen-based pharmacotherapies may be of value to women with opioid use disorder.
Keywords: Addiction; Estradiol; Opioid; Progesterone; Self-administration.
Conflict of interest statement
Figures
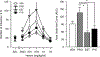
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

