A Novel Second-Generation EP2 Receptor Antagonist Reduces Neuroinflammation and Gliosis After Status Epilepticus in Rats
- PMID: 33410110
- PMCID: PMC8423966
- DOI: 10.1007/s13311-020-00969-5
A Novel Second-Generation EP2 Receptor Antagonist Reduces Neuroinflammation and Gliosis After Status Epilepticus in Rats
Abstract
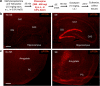
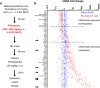
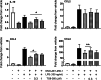
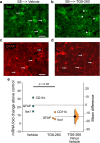
Prostaglandin-E2 (PGE2), an important mediator of inflammation, achieves its functions via four different G protein-coupled receptors (EP1, EP2, EP3, and EP4). We previously demonstrated that the EP2 receptor plays a proinflammatory and neurodegenerative role after status epilepticus (SE). We recently developed TG8-260 as a second-generation highly potent and selective EP2 antagonist. Here, we investigate whether TG8-260 is anti-inflammatory and combats neuropathology caused by pilocarpine-induced SE in rats. Adult male Sprague-Dawley rats were injected subcutaneously with pilocarpine (380-400 mg/kg) to induce SE. Following 60 min of SE, the rats were administered three doses of TG8-260 or vehicle and were allowed to recover. Neurodegeneration, neuroinflammation, gliosis, and blood-brain barrier (BBB) integrity were examined 4 days after SE. The results confirmed that pilocarpine-induced SE results in hippocampal neurodegeneration and a robust inflammatory response that persists days after SE. Furthermore, inhibition of the EP2 receptor by TG8-260 administered beginning 2 h after SE significantly reduced hippocampal neuroinflammation and gliosis but, in distinction to the earlier generation EP2 antagonist, did not mitigate neuronal injury or BBB breakdown. Thus, attenuation of neuroinflammation and gliosis is a common feature of EP2 inhibition following SE.
Keywords: BBB.; COX-2; EP2; PGE2; Pilocarpine; TG8-260; gliosis; hippocampus; inflammation; neurodegeneration; status epilepticus.
© 2021. The American Society for Experimental NeuroTherapeutics, Inc.
Figures





References
-
- Rojas A, Ganesh T, Lelutiu N, Gueorguieva P, Dingledine R. Inhibition of the prostaglandin EP2 receptor is neuroprotective and accelerates functional recovery in a rat model of organophosphorus induced status epilepticus. Neuropharmacology. 2015;93:15–27. doi: 10.1016/j.neuropharm.2015.01.017. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

