Adipose stem cells exhibit mechanical memory and reduce fibrotic contracture in a rat elbow injury model
- PMID: 33411380
- PMCID: PMC8745456
- DOI: 10.1096/fj.202001274R
Adipose stem cells exhibit mechanical memory and reduce fibrotic contracture in a rat elbow injury model
Abstract
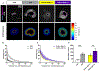
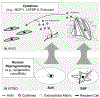
Fibrosis is driven by a misdirected cell response causing the overproduction of extracellular matrix and tissue dysfunction. Numerous pharmacological strategies have attempted to prevent fibrosis but have attained limited efficacy with some detrimental side effects. While stem cell treatments have provided more encouraging results, they have exhibited high variability and have not always improved tissue function. To enhance stem cell efficacy, we evaluated whether mechanical memory could direct cell response. We hypothesized that mechanically pre-conditioning on a soft matrix (soft priming) will delay adipose-derived stem cell (ASC) transition to a pro-fibrotic phenotype, expanding their regenerative potential, and improving healing in a complex tissue environment. Primary ASCs isolated from rat and human subcutaneous fat exhibited mechanical memory, demonstrated by a delayed cell response to stiffness following two weeks of soft priming including decreased cell area, actin coherency, and extracellular matrix production compared to cells on stiff substrates. Soft primed ASCs injected into our rat model of post-traumatic elbow contracture decreased histological evidence of anterior capsule fibrosis and increased elbow range-of-motion when evaluated by joint mechanics. These findings suggest that exploiting mechanical memory by strategically controlling the culture environment during cell expansion may improve the efficacy of stem cell-based therapies targeting fibrosis.
Keywords: anterior capsule; mechanical priming; post‐traumatic joint contracture; pre‐conditioning; range‐of‐motion.
© 2020 Federation of American Societies for Experimental Biology.
Figures


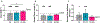
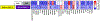

References
-
- Manka P, Zeller A, and Syn W-K (2019) Fibrosis in Chronic Liver Disease: An Update on Diagnostic and Treatment Modalities. Drugs 79, 903–927 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

