Prospective, historically controlled study to evaluate the efficacy and safety of a new paediatric formulation of nifurtimox in children aged 0 to 17 years with Chagas disease one year after treatment (CHICO)
- PMID: 33412557
- PMCID: PMC7790535
- DOI: 10.1371/journal.pntd.0008912
Prospective, historically controlled study to evaluate the efficacy and safety of a new paediatric formulation of nifurtimox in children aged 0 to 17 years with Chagas disease one year after treatment (CHICO)
Abstract
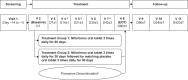
Nifurtimox is a recommended treatment for Chagas disease, but data from treated children are limited. We investigated the efficacy, safety and tolerability of nifurtimox administered as divisible, dispersible 30 mg and 120 mg tablets in children with Chagas disease. In this blinded, controlled study conducted January 2016-July 2018, 330 patients aged <18 years from 25 medical centres across three South American countries were randomised 2:1 to nifurtimox 10-20 mg/kg/day (aged <12 years) or 8-10 mg/kg/day (aged ≥12 years) for 60 days (n = 219), or for 30 days plus placebo for 30 days (n = 111) (ClinicalTrials.gov NCT02625974). The primary outcome was anti-Trypanosoma cruzi serological response (negative seroconversion or seroreduction ≥20% in mean optical density from baseline determined by two conventional enzyme-linked immunosorbent assays) at 12 months in the 60-day treatment group versus historical placebo controls. Nifurtimox for 60 days achieved negative seroconversion (n = 10) and seroreduction (n = 62) in 72 patients (serological response 32.9%; 95% confidence interval [CI] 26.4%, 39.3%, of all treated patients), confirming superiority relative to the upper 95% CI of 16% for controls. In patients aged <8 months, 10/12 treated for 60 days (83.3%) and 5/7 treated for 30 days (71.4%) achieved negative seroconversion. Overall serological response was lower for 30-day than for 60-day nifurtimox (between-treatment difference 14.0% [95% CI 3.7%, 24.2%]). The frequency of T. cruzi-positive quantitative polymerase chain reactions decreased substantially from baseline levels (60-day regimen 53.4% versus 1.4%; 30-day regimen 51.4% versus 4.5%). Study drug-related treatment-emergent adverse events (TEAEs), which were observed in 62 patients (28.3%) treated for 60 days and 29 patients (26.1%) treated for 30 days, were generally mild or moderate and resolved without sequelae; 4.2% of all TEAEs led to nifurtimox discontinuation. Age- and weight-adjusted nifurtimox for 60 days achieved a serological response at 12 months post-treatment that was superior to historical placebo, was well tolerated and had a favourable safety profile in children with Chagas disease. Although, at 1 year serological follow-up, efficacy of the shorter nifurtimox treatment was not comparable to the 60-day treatment regimen for the overall study population, further long-term follow-up of the patients will provide important information about the progress of serological conversion in children treated with nifurtimox, as well as the potential efficacy difference between the two regimens over time.
Conflict of interest statement
I have read the journal's policy and the authors of this manuscript have the following competing interests: UG is an employee of Bayer AG. EH is an employee of Bayer US LLC. GM received personal fees from Bayer during the conduct of the study.
Figures

Similar articles
-
Redefining the treatment of Chagas disease: a review of recent clinical and pharmacological data for a novel formulation of nifurtimox.PLoS Negl Trop Dis. 2025 Feb 25;19(2):e0012849. doi: 10.1371/journal.pntd.0012849. eCollection 2025 Feb. PLoS Negl Trop Dis. 2025. PMID: 39999088 Free PMC article. Review.
-
Efficacy and Safety of Nifurtimox in Pediatric Patients with Chagas Disease: Results at 4-Year Follow-Up in a Prospective, Historically Controlled Study (CHICO SECURE).Antimicrob Agents Chemother. 2023 Apr 18;67(4):e0119322. doi: 10.1128/aac.01193-22. Epub 2023 Mar 28. Antimicrob Agents Chemother. 2023. PMID: 36975790 Free PMC article. Clinical Trial.
-
ELISA F29 -A therapeutic efficacy biomarker in Chagas disease: Evaluation in pediatric patients treated with nifurtimox and followed for 4 years post-treatment.PLoS Negl Trop Dis. 2023 Jun 23;17(6):e0011440. doi: 10.1371/journal.pntd.0011440. eCollection 2023 Jun. PLoS Negl Trop Dis. 2023. PMID: 37352322 Free PMC article. Clinical Trial.
-
Nifurtimox versus benznidazole or placebo for asymptomatic Trypanosoma cruzi infection (Equivalence of Usual Interventions for Trypanosomiasis - EQUITY): study protocol for a randomised controlled trial.Trials. 2019 Jul 15;20(1):431. doi: 10.1186/s13063-019-3423-3. Trials. 2019. PMID: 31307503 Free PMC article.
-
Trypanocidal drugs for late-stage, symptomatic Chagas disease (Trypanosoma cruzi infection).Cochrane Database Syst Rev. 2020 Dec 11;12(12):CD004102. doi: 10.1002/14651858.CD004102.pub3. Cochrane Database Syst Rev. 2020. PMID: 33305846 Free PMC article.
Cited by
-
Efficacy of short-course treatment for prevention of congenital transmission of Chagas disease: A retrospective cohort study.PLoS Negl Trop Dis. 2024 Jan 22;18(1):e0011895. doi: 10.1371/journal.pntd.0011895. eCollection 2024 Jan. PLoS Negl Trop Dis. 2024. PMID: 38252673 Free PMC article.
-
Trypanosoma cruzi Secreted Cyclophilin TcCyP19 as an Early Marker for Trypanocidal Treatment Efficiency.Int J Mol Sci. 2023 Jul 25;24(15):11875. doi: 10.3390/ijms241511875. Int J Mol Sci. 2023. PMID: 37569250 Free PMC article.
-
Nifurtimox for Treatment of Chagas Disease in Pediatric Patients: the Challenges of Applying Pharmacokinetic-Pharmacodynamic Principles to Dose Finding.AAPS J. 2022 Aug 24;24(5):92. doi: 10.1208/s12248-022-00742-w. AAPS J. 2022. PMID: 36002604
-
Serological response to nifurtimox in adult patients with chronic Chagas disease: An observational comparative study in Argentina.PLoS Negl Trop Dis. 2021 Oct 4;15(10):e0009801. doi: 10.1371/journal.pntd.0009801. eCollection 2021 Oct. PLoS Negl Trop Dis. 2021. PMID: 34606501 Free PMC article.
-
Redefining the treatment of Chagas disease: a review of recent clinical and pharmacological data for a novel formulation of nifurtimox.PLoS Negl Trop Dis. 2025 Feb 25;19(2):e0012849. doi: 10.1371/journal.pntd.0012849. eCollection 2025 Feb. PLoS Negl Trop Dis. 2025. PMID: 39999088 Free PMC article. Review.
References
-
- World Health Organization. Chagas disease (American trypanosomiasis). Epidemiology. 2020. WHO. Available from: https://www.who.int/chagas/epidemiology/en/. Accessed August 5, 2020. 10.1111/tmi.13465 - DOI - PubMed
-
- Álvarez-Hernández D-A, Franyuti-Kelly G-A, Díaz-López-Silva R, González-Chávez A-M, González-Hermosillo-Cornejo D, Vázquez-López R. Chagas disease: current perspectives on a forgotten disease. Rev Med Hosp Gen Méx. 2018; 81:154–164.
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

