Cardiovascular Toxicity of Immune Checkpoint Inhibitors: Clinical Risk Factors
- PMID: 33415405
- PMCID: PMC7790474
- DOI: 10.1007/s11912-020-01002-w
Cardiovascular Toxicity of Immune Checkpoint Inhibitors: Clinical Risk Factors
Abstract
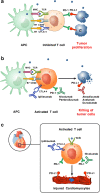
Purpose of review: Immune checkpoint inhibitors, such as monoclonal antibodies targeting CTLA-4, PD-1, and PD-L1, have improved the outcome of many malignancies, but serious immune-related cardiovascular adverse events have been observed. Patients' risk factors for these toxicities are currently being investigated.
Recent findings: Interfering with the CTLA-4 and PD-1 axes can bring to several immune-related adverse events, including cardiotoxic events such as autoimmune myocarditis, pericarditis, and vasculitis, suggesting that these molecules play an important role in preventing autoimmunity. Risk factors (such as pre-existing cardiovascular conditions, previous and concomitant cardiotoxic treatments, underlying autoimmune diseases, tumor-related factors, simultaneous immune-related toxic effects, and genetic factors) should be always recognized for the correct management of these toxicities.
Keywords: Cardio-immuno-oncology; Cardiotoxicity; Immune checkpoint inhibitors; Risk factors.
Conflict of interest statement
Flora Pirozzi, Remo Poto, Luisa Aran, Alessandra Cuomo, Giuseppe Spadaro, Pasquale Abete, Domenico Bonaduce, and Gilda Varricchi declare no conflict of interest. Maria Rosaria Galdiero is supported, in part, by a grant from the Italian Ministry of Education, University and Research (MIUR). Gianni Marone has received research funding through grants from the Regione Campania CISI-Lab, CRÈME Project, TIMING Project, and Federico II University. Carlo G. Tocchetti has received compensation from Alere for service as a consultant; is listed as an inventor on and receives royalties for Canadian Patent No. 2,613,477 (Thiol-Sensitive Positive Inotropes), issued 3 December 2013, owned by Johns Hopkins University (JHU Reference: C04755-P04755-05); and is listed as an inventor on U.S. Patent No. US20180271931 (P75NTR Antagonists and Treatment of Acute and Chronic Cardiac Disease), issued in 2018. Valentina Mercurio has received research funding from MSD.
Figures
References
-
- Tocchetti CG, Ameri P, de Boer RA, D’Alessandra Y, Russo M, Sorriento D, et al. Cardiac dysfunction in cancer patients: beyond direct cardiomyocyte damage of anticancer drugs. Novel cardio-oncology insights from the joint 2019 meeting of the ESC working groups of myocardial function and cellular biology of the heart. Cardiovasc Res. 2020;116(11):1820–1834. 10.1093/cvr/cvaa222. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

