Biological Characteristics and Osteogenic Differentiation of Ovine Bone Marrow Derived Mesenchymal Stem Cells Stimulated with FGF-2 and BMP-2
- PMID: 33419255
- PMCID: PMC7766718
- DOI: 10.3390/ijms21249726
Biological Characteristics and Osteogenic Differentiation of Ovine Bone Marrow Derived Mesenchymal Stem Cells Stimulated with FGF-2 and BMP-2
Abstract
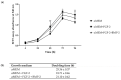
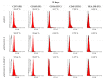
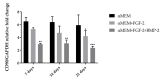
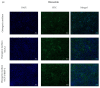
Cell-based therapies using mesenchymal stem cells (MSCs) are a promising tool in bone tissue engineering. Bone regeneration with MSCs involves a series of molecular processes leading to the activation of the osteoinductive cascade supported by bioactive factors, including fibroblast growth factor-2 (FGF-2) and bone morphogenetic protein-2 (BMP-2). In this study, we examined the biological characteristics and osteogenic differentiation potential of sheep bone marrow MSCs (BM-MSCs) treated with 20 ng/mL of FGF-2 and 100 ng/mL BMP-2 in vitro. The biological properties of osteogenic-induced BM-MSCs were investigated by assessing their morphology, proliferation, phenotype, and cytokine secretory profile. The osteogenic differentiation was characterized by Alizarin Red S staining, immunofluorescent staining of osteocalcin and collagen type I, and expression levels of genetic markers of osteogenesis. The results demonstrated that BM-MSCs treated with FGF-2 and BMP-2 maintained their primary MSC properties and improved their osteogenic differentiation capacity, as confirmed by increased expression of osteocalcin and collagen type I and upregulation of osteogenic-related gene markers BMP-2, Runx2, osterix, collagen type I, osteocalcin, and osteopontin. Furthermore, sheep BM-MSCs produced a variety of bioactive factors involved in osteogenesis, and supplementation of the culture medium with FGF-2 and BMP-2 affected the secretome profile of the cells. The results suggest that sheep osteogenic-induced BM-MSCs may be used as a cellular therapy to study bone repair in the preclinical large animal model.
Keywords: bone marrow MSCs; bone repair; large animal model; osteogenic differentiation.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures



Similar articles
-
Osteogenic Potential of Sheep Mesenchymal Stem Cells Preconditioned with BMP-2 and FGF-2 and Seeded on an nHAP-Coated PCL/HAP/β-TCP Scaffold.Cells. 2022 Oct 31;11(21):3446. doi: 10.3390/cells11213446. Cells. 2022. PMID: 36359842 Free PMC article.
-
[Effects of BMP-2 and FGF-2 on osteoblast differentiation of murine MSCs in vitro].Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2008 Nov;24(11):1062-5. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2008. PMID: 18992192 Chinese.
-
Gene Therapy for Bone Repair Using Human Cells: Superior Osteogenic Potential of Bone Morphogenetic Protein 2-Transduced Mesenchymal Stem Cells Derived from Adipose Tissue Compared to Bone Marrow.Hum Gene Ther. 2018 Apr;29(4):507-519. doi: 10.1089/hum.2017.097. Epub 2018 Mar 14. Hum Gene Ther. 2018. PMID: 29212377 Free PMC article.
-
Role of Adipose-Derived Mesenchymal Stem Cells in Bone Regeneration.Int J Mol Sci. 2024 Jun 20;25(12):6805. doi: 10.3390/ijms25126805. Int J Mol Sci. 2024. PMID: 38928517 Free PMC article. Review.
-
Rejuvenation of Bone Marrow Mesenchymal Stem Cells: Mechanisms and Their Application in Senile Osteoporosis Treatment.Biomolecules. 2025 Feb 13;15(2):276. doi: 10.3390/biom15020276. Biomolecules. 2025. PMID: 40001580 Free PMC article.
Cited by
-
Combination Effect of Rotator Cuff Repair with Secretome-hypoxia MSCs Ameliorates TNMD, RUNX2, and Healing Histology Score in Rotator Cuff Tear Rats.Arch Bone Jt Surg. 2023;11(10):617-624. doi: 10.22038/ABJS.2023.67933.3218. Arch Bone Jt Surg. 2023. PMID: 37873528 Free PMC article.
-
Promotion of Bone Formation in a Rat Osteoporotic Vertebral Body Defect Model via Suppression of Osteoclastogenesis by Ectopic Embryonic Calvaria Derived Mesenchymal Stem Cells.Int J Mol Sci. 2024 Jul 26;25(15):8174. doi: 10.3390/ijms25158174. Int J Mol Sci. 2024. PMID: 39125746 Free PMC article.
-
Image-based discrimination of the early stages of mesenchymal stem cell differentiation.Mol Biol Cell. 2024 Aug 1;35(8):ar103. doi: 10.1091/mbc.E24-02-0095. Epub 2024 Jun 5. Mol Biol Cell. 2024. PMID: 38837346 Free PMC article.
-
Human Non-Hypertrophic Nonunion Tissue Contains Osteoblast Lineage Cells and E-BMP-2 Activates Osteogenic and Chondrogenic Differentiation.Curr Issues Mol Biol. 2022 Nov 9;44(11):5562-5578. doi: 10.3390/cimb44110377. Curr Issues Mol Biol. 2022. PMID: 36354689 Free PMC article.
-
Mimicking Bone Extracellular Matrix: From BMP-2-Derived Sequences to Osteogenic-Multifunctional Coatings.Adv Healthc Mater. 2022 Oct;11(20):e2201339. doi: 10.1002/adhm.202201339. Epub 2022 Aug 15. Adv Healthc Mater. 2022. PMID: 35941083 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

