Resilin-mimetics as a smart biomaterial platform for biomedical applications
- PMID: 33420053
- PMCID: PMC7794388
- DOI: 10.1038/s41467-020-20375-x
Resilin-mimetics as a smart biomaterial platform for biomedical applications
Abstract
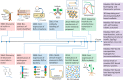
Intrinsically disordered proteins have dramatically changed the structure-function paradigm of proteins in the 21st century. Resilin is a native elastic insect protein, which features intrinsically disordered structure, unusual multi-stimuli responsiveness and outstanding resilience. Advances in computational techniques, polypeptide synthesis methods and modular protein engineering routines have led to the development of novel resilin-like polypeptides (RLPs) including modular RLPs, expanding their applications in tissue engineering, drug delivery, bioimaging, biosensors, catalysis and bioelectronics. However, how the responsive behaviour of RLPs is encoded in the amino acid sequence level remains elusive. This review summarises the milestones of RLPs, and discusses the development of modular RLP-based biomaterials, their current applications, challenges and future perspectives. A perspective of future research is that sequence and responsiveness profiling of RLPs can provide a new platform for the design and development of new modular RLP-based biomaterials with programmable structure, properties and functions.
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Chen Q, Liang S, Thouas GA. Elastomeric biomaterials for tissue engineering. Prog. Polym. Sci. 2013;38:584–671. doi: 10.1016/j.progpolymsci.2012.05.003. - DOI
-
- Weis-Fogh T. A rubber-like protein in insect cuticle. J. Exp. Biol. 1960;37:889.
-
- Elliott GF, Huxley AF, Weis-Fogh T. On the structure of resilin. J. Mol. Biol. 1965;13:791–795. doi: 10.1016/S0022-2836(65)80144-9. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

