An experimental pig model with outer retinal degeneration induced by temporary intravitreal loading of N-methyl-N-nitrosourea during vitrectomy
- PMID: 33420119
- PMCID: PMC7794530
- DOI: 10.1038/s41598-020-79437-1
An experimental pig model with outer retinal degeneration induced by temporary intravitreal loading of N-methyl-N-nitrosourea during vitrectomy
Abstract
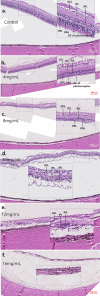
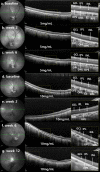
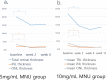
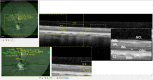
We aimed to develop an outer retinal degeneration pig model induced by temporary intravitreal loading of N-methyl-N-nitrosourea (MNU) during vitrectomy. In a preliminary experiment involving 5 mini-pig cases to determine the appropriate concentration of MNU, the vitreous cavity of each eye was filled with 4, 8, 10, 12, or 16 mg/mL MNU for 10 min, which was then replaced with a balanced salt solution. Multimodal examinations including spectral-domain optical coherence tomography (OCT) images and full-field electroretinography (ffERG) were obtained at baseline and week 2, week 6, and week 12. The retinal degeneration was classified according to the amplitudes of a dark adaptive (DA) 10.0 a-wave amplitude. The degree of moderate retinal degeneration was defined as DA 10.0 a-wave amplitude ≥ 10% and < 60% of baseline amplitude. The degree of severe degeneration was defined as DA 10.0 a-wave amplitude < 10% of baseline amplitude, noise, or flat signal. Hematoxylin and eosin staining and immunohistochemistry were performed at week 12. The main experiments were conducted first with 10 cases of 5 mg/mL and later with 13 cases of 10 mg/mL. In the preliminary experiment, degree of outer retinal degeneration increased with MNU concentration. Use of 4, 8, 10, 12, and 16 mg/mL MNU showed no, moderate, severe, severe, and atrophic changes, respectively. In the main experiments, there were 9 cases of moderate retinal degeneration and 1 case of severe degeneration in 5 mg/mL MNU group. Two cases of moderate degeneration and 11 of severe degeneration were recorded in 10 mg/mL group. Mean thickness of total retina, inner nuclear layer, and outer nuclear layer decreased at week 2 in both groups. The mean amplitudes on ffERG decreased at week 2. The ffERG and OCT findings did not change from week 2 to week 6 or week 12. The results of staining supported those of ffERG and OCT. Temporal MNU loading in a vitrectomized pig-eye model induced customized outer retinal degeneration with changing the concentration of MNU.
Conflict of interest statement
The authors declare no competing interests.
Figures






References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

