Sensitive detection of extremely small iron oxide nanoparticles in living mice using MP2RAGE with advanced image co-registration
- PMID: 33420210
- PMCID: PMC7794370
- DOI: 10.1038/s41598-020-80181-9
Sensitive detection of extremely small iron oxide nanoparticles in living mice using MP2RAGE with advanced image co-registration
Abstract
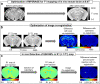
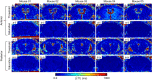
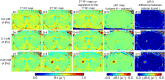
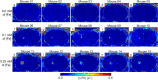
Magnetic resonance imaging (MRI) is a widely used non-invasive methodology for both preclinical and clinical studies. However, MRI lacks molecular specificity. Molecular contrast agents for MRI would be highly beneficial for detecting specific pathological lesions and quantitatively evaluating therapeutic efficacy in vivo. In this study, an optimized Magnetization Prepared-RApid Gradient Echo (MP-RAGE) with 2 inversion times called MP2RAGE combined with advanced image co-registration is presented as an effective non-invasive methodology to quantitatively detect T1 MR contrast agents. The optimized MP2RAGE produced high quality in vivo mouse brain T1 (or R1 = 1/T1) map with high spatial resolution, 160 × 160 × 160 µm3 voxel at 9.4 T. Test-retest signal to noise was > 20 for most voxels. Extremely small iron oxide nanoparticles (ESIONPs) having 3 nm core size and 11 nm hydrodynamic radius after polyethylene glycol (PEG) coating were intracranially injected into mouse brain and detected as a proof-of-concept. Two independent MP2RAGE MR scans were performed pre- and post-injection of ESIONPs followed by advanced image co-registration. The comparison of two T1 (or R1) maps after image co-registration provided precise and quantitative assessment of the effects of the injected ESIONPs at each voxel. The proposed MR protocol has potential for future use in the detection of T1 molecular contrast agents.
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Redox-triggered aggregation of ESIONPs with switchable T1 to T2 contrast effect for T2-weighted magnetic resonance imaging.J Mater Chem B. 2021 Feb 25;9(7):1821-1832. doi: 10.1039/d0tb02411b. J Mater Chem B. 2021. PMID: 33508067
-
Targeted MR Imaging Adopting T1-Weighted Ultra-Small Iron Oxide Nanoparticles for Early Hepatocellular Carcinoma: An in vitro and in vivo Study.Chin Med Sci J. 2020 Jun 30;35(2):142-150. doi: 10.24920/003643. Chin Med Sci J. 2020. PMID: 32684234
-
Extremely Small Iron Oxide Nanoparticle-Encapsulated Nanogels as a Glutathione-Responsive T1 Contrast Agent for Tumor-Targeted Magnetic Resonance Imaging.ACS Appl Mater Interfaces. 2020 Jun 17;12(24):26973-26981. doi: 10.1021/acsami.0c07288. Epub 2020 Jun 5. ACS Appl Mater Interfaces. 2020. PMID: 32452664
-
Hypoxia-Responsive T2-to-T1 Dynamically Switchable Extremely Small Iron Oxide Nanoparticles for Sensitive Tumor Imaging In Vivo.Bioconjug Chem. 2023 Sep 20;34(9):1622-1632. doi: 10.1021/acs.bioconjchem.3c00285. Epub 2023 Aug 16. Bioconjug Chem. 2023. PMID: 37584604
-
Optimization of MP2RAGE T1 mapping with radial view-ordering for deep brain stimulation targeting at 7 T MRI.Magn Reson Imaging. 2023 Jul;100:55-63. doi: 10.1016/j.mri.2023.03.007. Epub 2023 Mar 15. Magn Reson Imaging. 2023. PMID: 36924805
Cited by
-
Enhanced in vivo blood brain barrier transcytosis of macromolecular cargo using an engineered pH-sensitive mouse transferrin receptor binding nanobody.Fluids Barriers CNS. 2023 Aug 24;20(1):64. doi: 10.1186/s12987-023-00462-z. Fluids Barriers CNS. 2023. PMID: 37620930 Free PMC article.
-
Bioimaging guided pharmaceutical evaluations of nanomedicines for clinical translations.J Nanobiotechnology. 2022 May 19;20(1):236. doi: 10.1186/s12951-022-01451-4. J Nanobiotechnology. 2022. PMID: 35590412 Free PMC article. Review.
-
Selection of single domain anti-transferrin receptor antibodies for blood-brain barrier transcytosis using a neurotensin based assay and histological assessment of target engagement in a mouse model of Alzheimer's related amyloid-beta pathology.PLoS One. 2022 Oct 18;17(10):e0276107. doi: 10.1371/journal.pone.0276107. eCollection 2022. PLoS One. 2022. PMID: 36256604 Free PMC article.
-
Enhanced in Vivo Blood Brain Barrier Transcytosis of Macromolecular Cargo Using an Engineered pH-sensitive Mouse Transferrin Receptor Binding Nanobody.bioRxiv [Preprint]. 2023 Apr 27:2023.04.26.538462. doi: 10.1101/2023.04.26.538462. bioRxiv. 2023. Update in: Fluids Barriers CNS. 2023 Aug 24;20(1):64. doi: 10.1186/s12987-023-00462-z. PMID: 37333358 Free PMC article. Updated. Preprint.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

