Modeling the synergistic properties of drugs in hormonal treatment for prostate cancer
- PMID: 33422609
- PMCID: PMC7897323
- DOI: 10.1016/j.jtbi.2020.110570
Modeling the synergistic properties of drugs in hormonal treatment for prostate cancer
Abstract
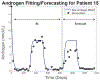
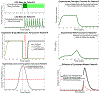
Prostate cancer is one of the most prevalent cancers in men, with increasing incidence worldwide. This public health concern has inspired considerable effort to study various aspects of prostate cancer treatment using dynamical models, especially in clinical settings. The standard of care for metastatic prostate cancer is hormonal therapy, which reduces the production of androgen that fuels the growth of prostate tumor cells prior to treatment resistance. Existing population models often use patients' prostate-specific antigen levels as a biomarker for model validation and for finding optimal treatment schedules; however, the synergistic effects of drugs used in hormonal therapy have not been well-examined. This paper describes the first mathematical model that explicitly incorporates the synergistic effects of two drugs used to inhibit androgen production in hormonal therapy. The drugs are cyproterone acetate, representing the drug family of anti-androgens that affect luteinizing hormones, and leuprolide acetate, representing the drug family of gonadotropin-releasing hormone analogs. By fitting the model to clinical data, we show that the proposed model can capture the dynamics of serum androgen levels during intermittent hormonal therapy better than previously published models. Our results highlight the importance of considering the synergistic effects of drugs in cancer treatment, thus suggesting that the dynamics of the drugs should be taken into account in optimal treatment studies, particularly for adaptive therapy. Otherwise, an unrealistic treatment schedule may be prescribed and render the treatment less effective. Furthermore, the drug dynamics allow our model to explain the delay in the relapse of androgen the moment a patient is taken off treatment, which supports that this delay is due to the residual effects of the drugs.
Keywords: Adaptive therapy; Androgen dynamics; Drug effects; Hormonal therapy; Intermittent androgen deprivation therapy; Optimal treatment schedule; Pharmacokinetics; Prostate cancer modeling.
Copyright © 2021 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Conflict of Interest
The authors declare no conflict of interest.
Figures





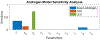

Similar articles
-
Intermittent androgen deprivation therapy in advanced prostate cancer.Curr Treat Options Oncol. 2014 Mar;15(1):127-36. doi: 10.1007/s11864-013-0272-2. Curr Treat Options Oncol. 2014. PMID: 24395278 Review.
-
Intermittent complete androgen blockade in PSA relapse after radical prostatectomy and incidental prostate cancer.Eur Urol. 1999;35 Suppl 1:27-31. Eur Urol. 1999. PMID: 10081700
-
The long-term results with delayed-combined androgen blockade therapy in local or locally advanced prostate cancer.Jpn J Clin Oncol. 2012 Jun;42(6):534-40. doi: 10.1093/jjco/hys037. Epub 2012 Mar 20. Jpn J Clin Oncol. 2012. PMID: 22438406
-
Final results of the Canadian prospective phase II trial of intermittent androgen suppression for men in biochemical recurrence after radiotherapy for locally advanced prostate cancer: clinical parameters.Cancer. 2006 Jul 15;107(2):389-95. doi: 10.1002/cncr.21989. Cancer. 2006. PMID: 16783817 Clinical Trial.
-
Androgen deprivation therapy with Leuprolide acetate for treatment of advanced prostate cancer.Expert Opin Pharmacother. 2017 Jan;18(1):105-113. doi: 10.1080/14656566.2016.1258058. Epub 2016 Nov 28. Expert Opin Pharmacother. 2017. PMID: 27826989 Review.
Cited by
-
Involvement of FAM83 Family Proteins in the Development of Solid Tumors: An Update Review.J Cancer. 2023 Jun 26;14(10):1888-1903. doi: 10.7150/jca.83420. eCollection 2023. J Cancer. 2023. PMID: 37476189 Free PMC article. Review.
-
High Accuracy Indicators of Androgen Suppression Therapy Failure for Prostate Cancer-A Modeling Study.Cancers (Basel). 2022 Aug 20;14(16):4033. doi: 10.3390/cancers14164033. Cancers (Basel). 2022. PMID: 36011026 Free PMC article.
-
Practical Understanding of Cancer Model Identifiability in Clinical Applications.Life (Basel). 2023 Feb 1;13(2):410. doi: 10.3390/life13020410. Life (Basel). 2023. PMID: 36836767 Free PMC article.
-
A Numerical Comparison of Petri Net and Ordinary Differential Equation SIR Component Models.ArXiv [Preprint]. 2025 Jul 1:arXiv:2407.10019v3. ArXiv. 2025. PMID: 39070030 Free PMC article. Preprint.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

