Novel immortalized human vocal fold epithelial cell line: In vitro tool for mucosal biology
- PMID: 33428261
- PMCID: PMC7839467
- DOI: 10.1096/fj.202001423R
Novel immortalized human vocal fold epithelial cell line: In vitro tool for mucosal biology
Erratum in
-
Erratum.FASEB J. 2022 Aug;36(8):e22439. doi: 10.1096/fsb2.22439. FASEB J. 2022. PMID: 35779043 Free PMC article. No abstract available.
Abstract
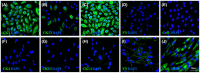
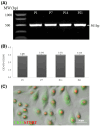
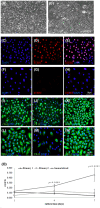
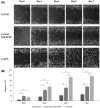
Study of vocal fold (VF) mucosal biology requires essential human vocal fold epithelial cell (hVFE) lines for use in appropriate model systems. We steadily transfected a retroviral construct containing human telomerase reverse transcriptase (hTERT) into primary normal hVFE to establish a continuously replicating hVFE cell line. Immortalized hVFE across passages have cobblestone morphology, express epithelial markers cytokeratin 4, 13 and 14, induced hTERT gene and protein expression, have similar RNAseq profiling, and can continuously grow for more than 8 months. DNA fingerprinting and karyotype analysis demonstrated that immortalized hVFE were consistent with the presence of a single cell line. Validation of the hVFE, in a three-dimensional in vitro VF mucosal construct revealed a multilayered epithelial structure with VF epithelial cell markers. Wound scratch assay revealed higher migration capability of the immortalized hVFE on the surface of collagen-fibronectin and collagen gel containing human vocal fold fibroblasts (hVFF). Collectively, our report demonstrates the first immortalized hVFE from true VFs providing a novel and invaluable tool for the study of epithelial cell-fibroblast interactions that dictate disease and health of this specialized tissue.
Keywords: human vocal fold epithelial cells; immortalization; telomerase; three-dimensional VF mucosal model.
© 2021 The Authors. The FASEB Journal published by Wiley Periodicals LLC on behalf of Federation of American Societies for Experimental Biology.
Conflict of interest statement
The authors have stated explicitly that there are no conflict of interest in connection with this article.
Figures




References
-
- Hirano M. Morphological structure of the vocal cord as a vibrator and its variations. Folia Phoniatr. 1974;26:89‐94. - PubMed
-
- Roy N, Merrill RM, Gray SD, Smith EM. Voice disorders in the general population: prevalence, risk factors, and occupational impact. Laryngoscope. 2005;115:1988‐1995. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

