Opportunity for Genotype-Guided Prescribing Among Adult Patients in 11 US Health Systems
- PMID: 33428770
- PMCID: PMC8217370
- DOI: 10.1002/cpt.2161
Opportunity for Genotype-Guided Prescribing Among Adult Patients in 11 US Health Systems
Abstract
The value of utilizing a multigene pharmacogenetic panel to tailor pharmacotherapy is contingent on the prevalence of prescribed medications with an actionable pharmacogenetic association. The Clinical Pharmacogenetics Implementation Consortium (CPIC) has categorized over 35 gene-drug pairs as "level A," for which there is sufficiently strong evidence to recommend that genetic information be used to guide drug prescribing. The opportunity to use genetic information to tailor pharmacotherapy among adult patients was determined by elucidating the exposure to CPIC level A drugs among 11 Implementing Genomics In Practice Network (IGNITE)-affiliated health systems across the US. Inpatient and/or outpatient electronic-prescribing data were collected between January 1, 2011 and December 31, 2016 for patients ≥ 18 years of age who had at least one medical encounter that was eligible for drug prescribing in a calendar year. A median of ~ 7.2 million adult patients was available for assessment of drug prescribing per year. From 2011 to 2016, the annual estimated prevalence of exposure to at least one CPIC level A drug prescribed to unique patients ranged between 15,719 (95% confidence interval (CI): 15,658-15,781) in 2011 to 17,335 (CI: 17,283-17,386) in 2016 per 100,000 patients. The estimated annual exposure to at least 2 drugs was above 7,200 per 100,000 patients in most years of the study, reaching an apex of 7,660 (CI: 7,632-7,687) per 100,000 patients in 2014. An estimated 4,748 per 100,000 prescribing events were potentially eligible for a genotype-guided intervention. Results from this study show that a significant portion of adults treated at medical institutions across the United States is exposed to medications for which genetic information, if available, should be used to guide prescribing.
© 2021 The Authors. Clinical Pharmacology & Therapeutics © 2021 American Society for Clinical Pharmacology and Therapeutics.
Conflict of interest statement
Figures
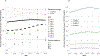


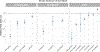
References
-
- Administration U.F.a.D. Table of pharmacogenomic biomarkers in drug labeling. (US Food and Drug Administration; ).
-
- Administration U.F.a.D. Table of Pharmacogenetic Associations. (US Food and Drug Administration; ).
Publication types
MeSH terms
Grants and funding
- U01 HG007269/HG/NHGRI NIH HHS/United States
- P30 CA076292/CA/NCI NIH HHS/United States
- UL1 TR001427/TR/NCATS NIH HHS/United States
- R01 HL092173/HL/NHLBI NIH HHS/United States
- U01 HG007762/HG/NHGRI NIH HHS/United States
- U01 HG007253/HG/NHGRI NIH HHS/United States
- UL1 TR000445/TR/NCATS NIH HHS/United States
- U01 HG010232/HG/NHGRI NIH HHS/United States
- UL1 TR000165/TR/NCATS NIH HHS/United States
- K24 HL133373/HL/NHLBI NIH HHS/United States
- U01 HG007775/HG/NHGRI NIH HHS/United States
- T32 HG008961/HG/NHGRI NIH HHS/United States
- U01 HG010245/HG/NHGRI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

