Efficacy and safety of the syrup "KalobaTUSS®" as a treatment for cough in children: a randomized, double blind, placebo-controlled clinical trial
- PMID: 33430841
- PMCID: PMC7798282
- DOI: 10.1186/s12887-020-02490-2
Efficacy and safety of the syrup "KalobaTUSS®" as a treatment for cough in children: a randomized, double blind, placebo-controlled clinical trial
Abstract
Background: Acute cough in children often causes discomfort to children and parents, reducing their quality of life. Despite the extensive utilization of over-the-counter remedies for cough, the efficacy of most of these treatments in children has not been confirmed.
Methods: We conducted a randomized, double blind, placebo-controlled clinical trial of 106 children with acute cough to evaluate the efficacy and safety of KalobaTUSS®, a paediatric cough syrup based on acacia honey and on Malva sylvestris extract, Inula helenium extract, Plantago major extract, and Helichrysum stoechas extract by using a validated 6 points Likert scale.
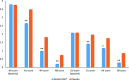
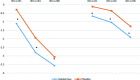
Results: Children were orally treated with KalobaTUSS® or placebo for 8 days. Children receiving KalobaTUSS® showed an early and significant reduction in night-time and day-time cough scores measured using a specific scale and a shorter duration of cough than children treated with the placebo.
Conclusions: KalobaTUSS® is well tolerated and produces positive effects by reducing the severity and shortening the duration of cough in children.
Trial registration: Clinicaltrials.gov no. NCT04073251 . Retrospectively registered.
Keywords: Children; Cough; Helichrysum stoechas; Honey; Inula helenium; Malva sylvestris; Plantago major; Syrup.
Conflict of interest statement
Authors Ilaria Carnevali, Lara Pauletto, Floriana Raso, Marco Testa, are employed by the company Schwabe Pharma Italia, Egna (BZ), Italy. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationship that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Efficacy of Grintuss® pediatric syrup in treating cough in children: a randomized, multicenter, double blind, placebo-controlled clinical trial.Ital J Pediatr. 2014 Jun 10;40:56. doi: 10.1186/1824-7288-40-56. Ital J Pediatr. 2014. PMID: 24917119 Free PMC article. Clinical Trial.
-
Efficacy and tolerability of a polysaccharide-resin-honey based cough syrup as compared to carbocysteine syrup for children with colds: a randomized, single-blinded, multicenter study.World J Pediatr. 2017 Feb;13(1):27-33. doi: 10.1007/s12519-016-0048-4. Epub 2016 Jul 15. World J Pediatr. 2017. PMID: 27457790 Clinical Trial.
-
Efficacy and safety of Chinese herbal medicine children's Zibei Xuanfei syrup in treating acute trachea-bronchitis with wind-heat invading lung syndrome: A randomized, double-blind, multicentre, controlled trial.J Ethnopharmacol. 2023 Jun 12;309:116207. doi: 10.1016/j.jep.2023.116207. Epub 2023 Feb 2. J Ethnopharmacol. 2023. PMID: 36736712 Clinical Trial.
-
Efficacy and tolerability of SEDIFLÙ in treating dry or productive cough in the pediatric population (SEPEDIA): A pilot, randomized, double-blind, placebo-controlled, multicenter clinical trial.Clin Pediatr (Phila). 2024 Nov;63(11):1510-1519. doi: 10.1177/00099228241228074. Epub 2024 Feb 7. Clin Pediatr (Phila). 2024. PMID: 38323572 Free PMC article. Clinical Trial.
-
Efficacy and tolerability of a fluid extract combination of thyme herb and ivy leaves and matched placebo in adults suffering from acute bronchitis with productive cough. A prospective, double-blind, placebo-controlled clinical trial.Arzneimittelforschung. 2006;56(9):652-60. doi: 10.1055/s-0031-1296767. Arzneimittelforschung. 2006. PMID: 17063641 Clinical Trial.
Cited by
-
Involvement of NO/cGMP Signaling Pathway, Ca2+ and K+ Channels on Spasmolytic Effect of Everlasting Flower Polyphenolic Extract (Helichrysum stoechas (L.) Moench).Int J Mol Sci. 2022 Nov 20;23(22):14422. doi: 10.3390/ijms232214422. Int J Mol Sci. 2022. PMID: 36430899 Free PMC article.
-
Effect of Plantago major on cough severity in acute bronchitis: A double-blind randomized clinical trial.Caspian J Intern Med. 2024 Sep 7;15(4):651-658. doi: 10.22088/cjim.15.4.651. eCollection 2024 Fall. Caspian J Intern Med. 2024. PMID: 39359439 Free PMC article.
-
Comparing the Effectiveness of Honey Consumption With Anti-Cough Medication in Pediatric Patients: A Systematic Review.Cureus. 2022 Sep 20;14(9):e29346. doi: 10.7759/cureus.29346. eCollection 2022 Sep. Cureus. 2022. PMID: 36284810 Free PMC article. Review.
-
Honey for acute cough in children - a systematic review.Eur J Pediatr. 2023 Sep;182(9):3949-3956. doi: 10.1007/s00431-023-05066-1. Epub 2023 Jun 25. Eur J Pediatr. 2023. PMID: 37355498 Free PMC article.
-
From Monographs to Chromatograms: The Antimicrobial Potential of Inula helenium L. (Elecampane) Naturalised in Ireland.Molecules. 2022 Feb 18;27(4):1406. doi: 10.3390/molecules27041406. Molecules. 2022. PMID: 35209195 Free PMC article.
References
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical