Roles of palmitoylation in structural long-term synaptic plasticity
- PMID: 33430908
- PMCID: PMC7802216
- DOI: 10.1186/s13041-020-00717-y
Roles of palmitoylation in structural long-term synaptic plasticity
Abstract
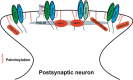
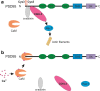
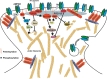
Long-term potentiation (LTP) and long-term depression (LTD) are important cellular mechanisms underlying learning and memory processes. N-Methyl-D-aspartate receptor (NMDAR)-dependent LTP and LTD play especially crucial roles in these functions, and their expression depends on changes in the number and single channel conductance of the major ionotropic glutamate receptor α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPAR) located on the postsynaptic membrane. Structural changes in dendritic spines comprise the morphological platform and support for molecular changes in the execution of synaptic plasticity and memory storage. At the molecular level, spine morphology is directly determined by actin cytoskeleton organization within the spine and indirectly stabilized and consolidated by scaffold proteins at the spine head. Palmitoylation, as a uniquely reversible lipid modification with the ability to regulate protein membrane localization and trafficking, plays significant roles in the structural and functional regulation of LTP and LTD. Altered structural plasticity of dendritic spines is also considered a hallmark of neurodevelopmental disorders, while genetic evidence strongly links abnormal brain function to impaired palmitoylation. Numerous studies have indicated that palmitoylation contributes to morphological spine modifications. In this review, we have gathered data showing that the regulatory proteins that modulate the actin network and scaffold proteins related to AMPAR-mediated neurotransmission also undergo palmitoylation and play roles in modifying spine architecture during structural plasticity.
Keywords: AMPAR; Actin cytoskeleton; Lipid posttranslational modification; Long term depression (LTD); Long term potentiation (LTP); Rho GTPases; Structural plasticity.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
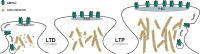

Similar articles
-
Regulation of neuronal PKA signaling through AKAP targeting dynamics.Eur J Cell Biol. 2006 Jul;85(7):627-33. doi: 10.1016/j.ejcb.2006.01.010. Epub 2006 Feb 28. Eur J Cell Biol. 2006. PMID: 16504338 Review.
-
Palmitoylation of A-kinase anchoring protein 79/150 regulates dendritic endosomal targeting and synaptic plasticity mechanisms.J Neurosci. 2012 May 23;32(21):7119-36. doi: 10.1523/JNEUROSCI.0784-12.2012. J Neurosci. 2012. PMID: 22623657 Free PMC article.
-
Role of Drebrin in Synaptic Plasticity.Adv Exp Med Biol. 2017;1006:183-201. doi: 10.1007/978-4-431-56550-5_11. Adv Exp Med Biol. 2017. PMID: 28865021 Review.
-
The N-methyl-D-aspartate receptor antagonist CPP alters synapse and spine structure and impairs long-term potentiation and long-term depression induced morphological plasticity in dentate gyrus of the awake rat.Neuroscience. 2010 Feb 17;165(4):1170-81. doi: 10.1016/j.neuroscience.2009.11.047. Epub 2009 Dec 1. Neuroscience. 2010. PMID: 19961908
-
Control of Homeostatic Synaptic Plasticity by AKAP-Anchored Kinase and Phosphatase Regulation of Ca2+-Permeable AMPA Receptors.J Neurosci. 2018 Mar 14;38(11):2863-2876. doi: 10.1523/JNEUROSCI.2362-17.2018. Epub 2018 Feb 13. J Neurosci. 2018. PMID: 29440558 Free PMC article.
Cited by
-
Lipids in Psychiatric Disorders: Functional and Potential Diagnostic Role as Blood Biomarkers.Metabolites. 2024 Jan 23;14(2):80. doi: 10.3390/metabo14020080. Metabolites. 2024. PMID: 38392971 Free PMC article. Review.
-
Treadmill exercise pretreatment ameliorated structural synaptic plasticity impairments of medial prefrontal cortex in vascular dementia rat and improved recognition memory.Sci Rep. 2024 Mar 26;14(1):7116. doi: 10.1038/s41598-024-57080-4. Sci Rep. 2024. PMID: 38531892 Free PMC article.
-
Stress resilience is an active and multifactorial process manifested by structural, functional, and molecular changes in synapses.Neurobiol Stress. 2024 Oct 22;33:100683. doi: 10.1016/j.ynstr.2024.100683. eCollection 2024 Nov. Neurobiol Stress. 2024. PMID: 39524934 Free PMC article.
-
Altered Cortical Palmitoylation Induces Widespread Molecular Disturbances in Parkinson's Disease.Int J Mol Sci. 2022 Nov 14;23(22):14018. doi: 10.3390/ijms232214018. Int J Mol Sci. 2022. PMID: 36430497 Free PMC article.
-
Proteomic analysis of s-acylated proteins in human retinal pigment epithelial cells and the role of palmitoylation of Niemann-Pick type C1 protein in cholesterol transport.Front Aging Neurosci. 2022 Oct 3;14:965943. doi: 10.3389/fnagi.2022.965943. eCollection 2022. Front Aging Neurosci. 2022. PMID: 36262888 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

