Improving preclinical studies through replications
- PMID: 33432925
- PMCID: PMC7817176
- DOI: 10.7554/eLife.62101
Improving preclinical studies through replications
Abstract
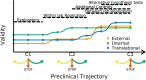
The purpose of preclinical research is to inform the development of novel diagnostics or therapeutics, and the results of experiments on animal models of disease often inform the decision to conduct studies in humans. However, a substantial number of clinical trials fail, even when preclinical studies have apparently demonstrated the efficacy of a given intervention. A number of large-scale replication studies are currently trying to identify the factors that influence the robustness of preclinical research. Here, we discuss replications in the context of preclinical research trajectories, and argue that increasing validity should be a priority when selecting experiments to replicate and when performing the replication. We conclude that systematically improving three domains of validity - internal, external and translational - will result in a more efficient allocation of resources, will be more ethical, and will ultimately increase the chances of successful translation.
Keywords: medicine; none; preclinical research; replication; reproducibility; science forum; translation; validity.
© 2021, Drude et al.
Conflict of interest statement
ND, LM, MD, UD, UT No competing interests declared
Figures
References
-
- Albers C, Lakens D. When power analyses based on pilot data are biased: inaccurate effect size estimators and follow-up bias. Journal of Experimental Social Psychology. 2018;74:187–195. doi: 10.1016/j.jesp.2017.09.004. - DOI
-
- BMBF-DLR Confirmatory preclinical studies (Förderung von konfirmatorischen präklinischen studien) [January 14, 2021];German Federal Ministry of Education and Research. 2018 https://www.gesundheitsforschung-bmbf.de/de/8344.php
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

