Potential Suppressive Effect of Nicotine on the Inflammatory Response in Oral Epithelial Cells: An In Vitro Study
- PMID: 33435295
- PMCID: PMC7826768
- DOI: 10.3390/ijerph18020483
Potential Suppressive Effect of Nicotine on the Inflammatory Response in Oral Epithelial Cells: An In Vitro Study
Abstract
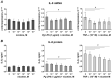
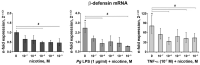
Smoking is a well-recognized risk factor for oral mucosal and periodontal diseases. Nicotine is an important component of cigarette smoke. This study aims to investigate the impact of nicotine on the viability and inflammatory mediator production of an oral epithelial cell line in the presence of various inflammatory stimuli. Oral epithelial HSC-2 cells were challenged with nicotine (10-8-10-2 M) for 24 h in the presence or absence of Porphyromonas gingivalis lipopolysaccharide (LPS, 1 µg/mL) or tumor necrosis factor (TNF)-α (10-7 M) for 24 h. The cell proliferation/viability was determined by MTT assay. Gene expression of interleukin (IL)-8, intercellular adhesion molecule (ICAM)-1, and β-defensin was assayed by qPCR. The production of IL-8 protein and cell surface expression of ICAM-1 was assessed by ELISA and flow cytometry, respectively. Proliferation/viability of HSC-2 cells was unaffected by nicotine at concentrations up to 10-3 M and inhibited at 10-2 M. Nicotine had no significant effect on the basal expression of IL-8, ICAM-1, and β-defensin. At the same time, it significantly diminished P. gingivalis LPS or the TNF-α-induced expression levels of these factors. Within the limitations of this study, the first evidence was provided in vitro that nicotine probably exerts a suppressive effect on the production of inflammatory mediators and antimicrobial peptides in human oral epithelial cells.
Keywords: epithelial cell; inflammation; lipopolysaccharide; nicotine.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

