Generation of proliferating human adult hepatocytes using optimized 3D culture conditions
- PMID: 33436872
- PMCID: PMC7804446
- DOI: 10.1038/s41598-020-80019-4
Generation of proliferating human adult hepatocytes using optimized 3D culture conditions
Abstract
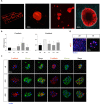
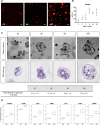
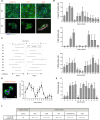
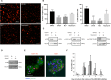
Generating the proliferation of differentiated normal adult human hepatocytes is a major challenge and an expected central step in understanding the microenvironmental conditions that regulate the phenotype of human hepatocytes in vitro. In this work, we described optimized 3D culture conditions of primary human hepatocytes (PHH) to trigger two waves of proliferation and we identified matrix stiffness and cell-cell interactions as the main actors necessary for this proliferation. We demonstrated that DNA replication and overexpression of cell cycle markers are modulate by the matrix stiffness while PHH cultured in 3D without prior cellular interactions did not proliferate. Besides, we showed that PHH carry out an additional cell cycle after transient inhibition of MAPK MER1/2-ERK1/2 signaling pathway. Collagen cultured hepatocytes are organized as characteristic hollow spheroids able to maintain survival, cell polarity and hepatic differentiation for long-term culture periods of at least 28 days. Remarkably, we demonstrated by transcriptomic analysis and functional experiments that proliferating cells are mature hepatocytes with high detoxication capacities. In conclusion, the advanced 3D model described here, named Hepoid, is particularly relevant for obtaining normal human proliferating hepatocytes. By allowing concomitant proliferation and differentiation, it constitutes a promising tool for many pharmacological and biotechnological applications.
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Increasing 3D Matrix Rigidity Strengthens Proliferation and Spheroid Development of Human Liver Cells in a Constant Growth Factor Environment.J Cell Biochem. 2016 Mar;117(3):708-20. doi: 10.1002/jcb.25356. Epub 2015 Sep 8. J Cell Biochem. 2016. PMID: 26331987
-
Three-Dimensional Spheroid Primary Human Hepatocytes in Monoculture and Coculture with Nonparenchymal Cells.Tissue Eng Part C Methods. 2018 Sep;24(9):534-545. doi: 10.1089/ten.TEC.2018.0134. Tissue Eng Part C Methods. 2018. PMID: 30101670
-
Direct self-assembly of hepatocytes spheroids within hollow fibers in presence of collagen.Biotechnol Lett. 2006 Feb;28(4):279-84. doi: 10.1007/s10529-005-5531-2. Biotechnol Lett. 2006. PMID: 16555013
-
Three-Dimensional Liver Culture Systems to Maintain Primary Hepatic Properties for Toxicological Analysis In Vitro.Int J Mol Sci. 2021 Sep 23;22(19):10214. doi: 10.3390/ijms221910214. Int J Mol Sci. 2021. PMID: 34638555 Free PMC article. Review.
-
Advanced Techniques and Awaited Clinical Applications for Human Pluripotent Stem Cell Differentiation into Hepatocytes.Hepatology. 2021 Aug;74(2):1101-1116. doi: 10.1002/hep.31705. Epub 2021 Aug 22. Hepatology. 2021. PMID: 33420753 Free PMC article. Review.
Cited by
-
Primary-like Human Hepatocytes Genetically Engineered to Obtain Proliferation Competence as a Capable Application for Energy Metabolism Experiments in In Vitro Oncologic Liver Models.Biology (Basel). 2022 Aug 9;11(8):1195. doi: 10.3390/biology11081195. Biology (Basel). 2022. PMID: 36009822 Free PMC article.
-
A Preliminary Investigation of Embedding In Vitro HepaRG Spheroids into Recombinant Human Collagen Type I for the Promotion of Liver Differentiation.Polymers (Basel). 2022 May 9;14(9):1923. doi: 10.3390/polym14091923. Polymers (Basel). 2022. PMID: 35567092 Free PMC article.
-
From NAFLD to MAFLD: Aligning Translational In Vitro Research to Clinical Insights.Biomedicines. 2022 Jan 12;10(1):161. doi: 10.3390/biomedicines10010161. Biomedicines. 2022. PMID: 35052840 Free PMC article. Review.
-
Chronotoxici-Plate Containing Droplet-Engineered Rhythmic Liver Organoids for Drug Toxicity Evaluation.Adv Sci (Weinh). 2024 Jul;11(28):e2305925. doi: 10.1002/advs.202305925. Epub 2024 May 8. Adv Sci (Weinh). 2024. PMID: 38720476 Free PMC article.
-
Three-dimensional human bile duct formation from chemically induced human liver progenitor cells.Front Bioeng Biotechnol. 2023 Aug 21;11:1249769. doi: 10.3389/fbioe.2023.1249769. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 37671190 Free PMC article.
References
-
- Thoresen GH, Guren TK, Christoffersen T. Role of ERK, p38 and PI3-kinase in EGF receptor-mediated mitogenic signalling in cultured rat hepatocytes: Requirement for sustained ERK activation. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2003;13:229–238. doi: 10.1159/000072426. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

