Myelin-specific T cells in animals with Japanese macaque encephalomyelitis
- PMID: 33440071
- PMCID: PMC7886046
- DOI: 10.1002/acn3.51303
Myelin-specific T cells in animals with Japanese macaque encephalomyelitis
Abstract
Objective: To determine whether animals with Japanese macaque encephalomyelitis (JME), a spontaneous demyelinating disease similar to multiple sclerosis (MS), harbor myelin-specific T cells in their central nervous system (CNS) and periphery.
Methods: Mononuclear cells (MNCs) from CNS lesions, cervical lymph nodes (LNs) and peripheral blood of Japanese macaques (JMs) with JME, and cervical LN and blood MNCs from healthy controls or animals with non-JME conditions were analyzed for the presence of myelin-specific T cells and changes in interleukin 17 (IL-17) and interferon gamma (IFNγ) expression.
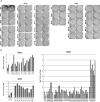
Results: Demyelinating JME lesions contained CD4+ T cells and CD8+ T cells specific to myelin oligodendrocyte glycoprotein (MOG), myelin basic protein (MBP), and/or proteolipid protein (PLP). CD8+ T-cell responses were absent in JME peripheral blood, and in age- and sex-matched controls. However, CD4+ Th1 and Th17 responses were detected in JME peripheral blood versus controls. Cervical LN MNCs from eight of nine JME animals had CD3+ T cells specific for MOG, MBP, and PLP that were not detected in controls. Mapping myelin epitopes revealed a heterogeneity in responses among JME animals. Comparison of myelin antigen sequences with those of JM rhadinovirus (JMRV), which is found in JME lesions, identified six viral open reading frames (ORFs) with similarities to myelin antigen sequences. Overlapping peptides to these JMRV ORFs did not induce IFNγ responses.
Interpretations: JME possesses an immune-mediated component that involves both CD4+ and CD8+ T cells specific for myelin antigens. JME may shed new light on inflammatory demyelinating disease pathogenesis linked to gamma-herpesvirus infection.
© 2021 The Authors. Annals of Clinical and Translational Neurology published by Wiley Periodicals LLC on behalf of American Neurological Association.
Conflict of interest statement
All authors have nothing to report.
Figures



References
-
- Peng Z, Zhang L, Wang H, et al. Experimental autoimmune encephalomyelitis (EAE) model of cynmolgus macaques induced by recombinant human MOB1‐125(rhMOG1‐125) protein and MOG34‐56 peptide. Protein Pept Lett 2017;24:1166–1178. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
