Hyaluronan-Loaded Liposomal Dexamethasone-Diclofenac Nanoparticles for Local Osteoarthritis Treatment
- PMID: 33440880
- PMCID: PMC7826786
- DOI: 10.3390/ijms22020665
Hyaluronan-Loaded Liposomal Dexamethasone-Diclofenac Nanoparticles for Local Osteoarthritis Treatment
Abstract
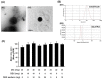
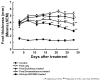
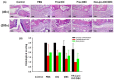
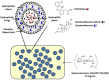
Osteoarthritis (OA) remains one of the common degenerative joint diseases and a major cause of pain and disability in older adult individuals. Oral administration of non-steroidal anti-inflammatory drugs (NSAIDs) (such as diclofenac, DIC) or intra-articular injected gluco-corticosteroids (such as dexamethasone, DEX) were the conventional treatment strategies for OA to reduce joint pain. Current limitations for both drugs including severe adverse effects with risks of toxicity were noted. The aim of the present study was to generate a novel OA treatment formulation hyaluronic acid (HA)-Liposomal (Lipo)-DIC/DEX to combat joint pain. The formulation was prepared by constructing DIC with DEX-loaded nanostructured lipid carriers Lipo-DIC/DEX mixed with hyaluronic acid (HA) for prolonged OA application. The prepared Lipo-DIC/DEX nanoparticles revealed the size as 103.6 ± 0.3 nm on average, zeta potential as -22.3 ± 4.6 mV, the entrapment efficiency of 90.5 ± 5.6%, and the DIC and DEX content was 22.5 ± 4.1 and 2.5 ± 0.6%, respectively. Evidence indicated that HA-Lipo-DIC/DEX could reach the effective working concentration in 4 h and sustained the drug-releasing time for at least 168 h. No significant toxicities but increased cell numbers were observed when HA-Lipo-DIC/DEX co-cultured with articular chondrocytes cells. Using live-animal In vivo imaging system (IVIS), intra-articular injection of each HA-Lipo-DIC/DEX sufficed to reduce knee joint inflammation in OA mice over a time span of four weeks. Single-dose injection could reduce the inflammation volume down to 77.5 ± 5.1% from initial over that time span. Our results provided the novel drug-releasing formulation with safety and efficiency which could be a promising system for osteoarthritis pain control.
Keywords: dexamethasone; diclofenac; liposomal nanoparticle; osteoarthritis.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures



Similar articles
-
Liposomal dexamethasone-moxifloxacin nanoparticle combinations with collagen/gelatin/alginate hydrogel for corneal infection treatment and wound healing.Biomed Mater. 2020 Aug 10;15(5):055022. doi: 10.1088/1748-605X/ab9510. Biomed Mater. 2020. PMID: 32434164
-
Liposomal dexamethasone-diclofenac combinations for local osteoarthritis treatment.Int J Pharm. 2009 Jul 6;376(1-2):84-91. doi: 10.1016/j.ijpharm.2009.04.025. Epub 2009 May 3. Int J Pharm. 2009. PMID: 19409466
-
Harnessing hyaluronic acid-based nanoparticles for combination therapy: A novel approach for suppressing systemic inflammation and to promote antitumor macrophage polarization.Carbohydr Polym. 2021 Feb 15;254:117291. doi: 10.1016/j.carbpol.2020.117291. Epub 2020 Oct 27. Carbohydr Polym. 2021. PMID: 33357860
-
Improving dexamethasone drug loading and efficacy in treating rheumatoid arthritis via liposome: Focusing on inflammation and molecular mechanisms.Animal Model Exp Med. 2025 Jan;8(1):5-19. doi: 10.1002/ame2.12518. Epub 2024 Dec 3. Animal Model Exp Med. 2025. PMID: 39627850 Free PMC article. Review.
-
Potential mechanism of action of intra-articular hyaluronan therapy in osteoarthritis: are the effects molecular weight dependent?Semin Arthritis Rheum. 2002 Aug;32(1):10-37. doi: 10.1053/sarh.2002.33720. Semin Arthritis Rheum. 2002. PMID: 12219318 Review.
Cited by
-
An update on the advances in the field of nanostructured drug delivery systems for a variety of orthopedic applications.Drug Deliv. 2023 Dec;30(1):2241667. doi: 10.1080/10717544.2023.2241667. Epub 2023 Dec 1. Drug Deliv. 2023. PMID: 38037335 Free PMC article.
-
Functional biomaterials for osteoarthritis treatment: From research to application.Smart Med. 2022 Dec 27;1(1):e20220014. doi: 10.1002/SMMD.20220014. eCollection 2022 Dec. Smart Med. 2022. PMID: 39188730 Free PMC article. Review.
-
Advances in Lipid-Based Codelivery Systems for Cancer and Inflammatory Diseases.Adv Healthc Mater. 2023 Mar;12(7):e2202400. doi: 10.1002/adhm.202202400. Epub 2022 Dec 16. Adv Healthc Mater. 2023. PMID: 36453542 Free PMC article. Review.
-
Subcutaneous toxicity of a dual ionically cross-linked atelocollagen and sodium hyaluronate gel: Rat in vivo study for biological safety evaluation of the injectable hydrogel.Toxicol Rep. 2021 Sep 13;8:1651-1656. doi: 10.1016/j.toxrep.2021.09.001. eCollection 2021. Toxicol Rep. 2021. PMID: 34567979 Free PMC article.
-
Advancing drug delivery to articular cartilage: From single to multiple strategies.Acta Pharm Sin B. 2023 Oct;13(10):4127-4148. doi: 10.1016/j.apsb.2022.11.021. Epub 2022 Nov 25. Acta Pharm Sin B. 2023. PMID: 37799383 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

