Extracellular Metabolism Sets the Table for Microbial Cross-Feeding
- PMID: 33441489
- PMCID: PMC7849352
- DOI: 10.1128/MMBR.00135-20
Extracellular Metabolism Sets the Table for Microbial Cross-Feeding
Abstract
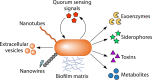
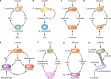
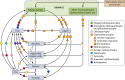
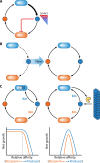
The transfer of nutrients between cells, or cross-feeding, is a ubiquitous feature of microbial communities with emergent properties that influence our health and orchestrate global biogeochemical cycles. Cross-feeding inevitably involves the externalization of molecules. Some of these molecules directly serve as cross-fed nutrients, while others can facilitate cross-feeding. Altogether, externalized molecules that promote cross-feeding are diverse in structure, ranging from small molecules to macromolecules. The functions of these molecules are equally diverse, encompassing waste products, enzymes, toxins, signaling molecules, biofilm components, and nutrients of high value to most microbes, including the producer cell. As diverse as the externalized and transferred molecules are the cross-feeding relationships that can be derived from them. Many cross-feeding relationships can be summarized as cooperative but are also subject to exploitation. Even those relationships that appear to be cooperative exhibit some level of competition between partners. In this review, we summarize the major types of actively secreted, passively excreted, and directly transferred molecules that either form the basis of cross-feeding relationships or facilitate them. Drawing on examples from both natural and synthetic communities, we explore how the interplay between microbial physiology, environmental parameters, and the diverse functional attributes of extracellular molecules can influence cross-feeding dynamics. Though microbial cross-feeding interactions represent a burgeoning field of interest, we may have only begun to scratch the surface.
Keywords: biofilm; coculture; cross-feeding; exoenzymes; microbial ecology; mutualism; nanowires; quorum sensing; siderophores; synthetic ecology.
Copyright © 2021 American Society for Microbiology.
Figures
Similar articles
-
Recipient-Biased Competition for an Intracellularly Generated Cross-Fed Nutrient Is Required for Coexistence of Microbial Mutualists.mBio. 2017 Nov 28;8(6):e01620-17. doi: 10.1128/mBio.01620-17. mBio. 2017. PMID: 29184014 Free PMC article.
-
Covert Cross-Feeding Revealed by Genome-Wide Analysis of Fitness Determinants in a Synthetic Bacterial Mutualism.Appl Environ Microbiol. 2020 Jun 17;86(13):e00543-20. doi: 10.1128/AEM.00543-20. Print 2020 Jun 17. Appl Environ Microbiol. 2020. PMID: 32332139 Free PMC article.
-
Quorum Sensing and Antimicrobial Production Orchestrate Biofilm Dynamics in Multispecies Bacterial Communities.Microbiol Spectr. 2022 Dec 21;10(6):e0261522. doi: 10.1128/spectrum.02615-22. Epub 2022 Oct 18. Microbiol Spectr. 2022. PMID: 36255295 Free PMC article.
-
Microbial biofilm ecology, in silico study of quorum sensing receptor-ligand interactions and biofilm mediated bioremediation.Arch Microbiol. 2021 Jan;203(1):13-30. doi: 10.1007/s00203-020-02012-9. Epub 2020 Aug 12. Arch Microbiol. 2021. PMID: 32785735 Review.
-
Dynamics in the mixed microbial concourse.Genes Dev. 2010 Dec 1;24(23):2603-14. doi: 10.1101/gad.1985210. Genes Dev. 2010. PMID: 21123647 Free PMC article. Review.
Cited by
-
Comparative Population Genetics in the Human Gut Microbiome.Genome Biol Evol. 2022 Jan 4;14(1):evab116. doi: 10.1093/gbe/evab116. Genome Biol Evol. 2022. PMID: 34028530 Free PMC article.
-
Increased privatization of a public resource leads to spread of cooperation in a microbial population.Microbiol Spectr. 2024 Feb 6;12(2):e0235823. doi: 10.1128/spectrum.02358-23. Epub 2024 Jan 11. Microbiol Spectr. 2024. PMID: 38206031 Free PMC article.
-
The Schizosaccharomyces pombe ornithine-N5-oxygenase Sib2 interacts with the N5-transacetylase Sib3 in the ferrichrome biosynthetic pathway.Front Microbiol. 2024 Sep 10;15:1467397. doi: 10.3389/fmicb.2024.1467397. eCollection 2024. Front Microbiol. 2024. PMID: 39328910 Free PMC article.
-
Profiling rhythmicity of bile salt hydrolase activity in the gut lumen with a rapid fluorescence assay.Org Biomol Chem. 2023 May 17;21(19):4028-4038. doi: 10.1039/d2ob02257e. Org Biomol Chem. 2023. PMID: 36810586 Free PMC article.
-
Quinolone-mediated metabolic cross-feeding develops aluminium tolerance in soil microbial consortia.Nat Commun. 2024 Nov 22;15(1):10148. doi: 10.1038/s41467-024-54616-0. Nat Commun. 2024. PMID: 39578460 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

