Local immune response to food antigens drives meal-induced abdominal pain
- PMID: 33442055
- PMCID: PMC7610810
- DOI: 10.1038/s41586-020-03118-2
Local immune response to food antigens drives meal-induced abdominal pain
Abstract
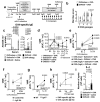
Up to 20% of people worldwide develop gastrointestinal symptoms following a meal1, leading to decreased quality of life, substantial morbidity and high medical costs. Although the interest of both the scientific and lay communities in this issue has increased markedly in recent years, with the worldwide introduction of gluten-free and other diets, the underlying mechanisms of food-induced abdominal complaints remain largely unknown. Here we show that a bacterial infection and bacterial toxins can trigger an immune response that leads to the production of dietary-antigen-specific IgE antibodies in mice, which are limited to the intestine. Following subsequent oral ingestion of the respective dietary antigen, an IgE- and mast-cell-dependent mechanism induced increased visceral pain. This aberrant pain signalling resulted from histamine receptor H1-mediated sensitization of visceral afferents. Moreover, injection of food antigens (gluten, wheat, soy and milk) into the rectosigmoid mucosa of patients with irritable bowel syndrome induced local oedema and mast cell activation. Our results identify and characterize a peripheral mechanism that underlies food-induced abdominal pain, thereby creating new possibilities for the treatment of irritable bowel syndrome and related abdominal pain disorders.
Conflict of interest statement
The authors declare no competing interests
Figures






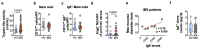




Comment in
-
Food for thought about the immune drivers of gut pain.Nature. 2021 Feb;590(7844):41-43. doi: 10.1038/d41586-020-03661-y. Nature. 2021. PMID: 33441989 No abstract available.
-
Understanding the immune drivers of food-induced abdominal pain.Nat Rev Gastroenterol Hepatol. 2021 Mar;18(3):149. doi: 10.1038/s41575-021-00422-8. Nat Rev Gastroenterol Hepatol. 2021. PMID: 33526865 No abstract available.
References
-
- Schäfer T, et al. Epidemiology of food allergy/food intolerance in adults: Associations with other manifestations of atopy. Allergy Eur J Allergy Clin Immunol. 2001;56:1172–1179. - PubMed
-
- Honda K, Littman DR. The microbiota in adaptive immune homeostasis and disease. Nature. 2016;535:75–84. - PubMed
-
- Mowat AMI. Anatomical basis of tolerance and immunity to intestinal antigens. Nature Reviews Immunology. 2003;3:331–341. - PubMed
-
- Spiller R, Garsed K. Postinfectious irritable bowel syndrome. Gastroenterology. 2009;136:1979–1988. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

