The interaction between brain and liver regulates lipid metabolism in the TBI pathology
- PMID: 33444711
- PMCID: PMC7889047
- DOI: 10.1016/j.bbadis.2021.166078
The interaction between brain and liver regulates lipid metabolism in the TBI pathology
Abstract
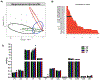
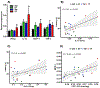
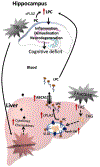
To shed light on the impact of systemic physiology on the pathology of traumatic brain injury (TBI), we examine the effects of TBI (concussive injury) and dietary fructose on critical aspects of lipid homeostasis in the brain and liver of young-adult rats. Lipids are integral components of brain structure and function, and the liver has a role on the synthesis and metabolism of lipids. Fructose is mainly metabolized in the liver with potential implications for brain function. Lipidomic analysis accompanied by unbiased sparse partial least squares discriminant analysis (sPLS-DA) identified lysophosphatidylcholine (LPC) and cholesterol ester (CE) as the top lipid families impacted by TBI and fructose in the hippocampus, and only LPC (16:0) was associated with hippocampal-dependent memory performance. Fructose and TBI elevated liver pro-inflammatory markers, interleukin-1α (IL-1α), Interferon-γ (IFN-γ) that correlated with hippocampal-dependent memory dysfunction, and monocyte chemoattractant protein-1 (MCP-1) positively correlated with LPC levels in the hippocampus. The effects of fructose were more pronounced in the liver, in agreement with the role of liver on fructose metabolism and suggest that fructose could exacerbate liver inflammation caused by TBI. The overall results indicate that TBI and fructose interact to influence systemic and central inflammation by engaging liver lipids. The impact of TBI and fructose diet on the periphery provides a therapeutic target to counteract the TBI pathogenesis.
Keywords: Cognition; Fructose; Liver; Lysophosphatidylcholine; Traumatic brain injury; cPLA2.
Copyright © 2021 Elsevier B.V. All rights reserved.
Conflict of interest statement
Competing Interests Statement
The authors declare no competing interests.
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
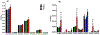
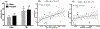
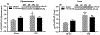
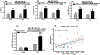
Similar articles
-
Brain Trauma Disrupts Hepatic Lipid Metabolism: Blame It on Fructose?Mol Nutr Food Res. 2019 Aug;63(15):e1801054. doi: 10.1002/mnfr.201801054. Epub 2019 Jun 6. Mol Nutr Food Res. 2019. PMID: 31087499 Free PMC article.
-
Liver acts as a metabolic gate for the traumatic brain injury pathology: Protective action of thyroid hormone.Biochim Biophys Acta Mol Basis Dis. 2023 Aug;1869(6):166728. doi: 10.1016/j.bbadis.2023.166728. Epub 2023 May 1. Biochim Biophys Acta Mol Basis Dis. 2023. PMID: 37137432 Free PMC article.
-
Dietary fructose aggravates the pathobiology of traumatic brain injury by influencing energy homeostasis and plasticity.J Cereb Blood Flow Metab. 2016 May;36(5):941-53. doi: 10.1177/0271678X15606719. Epub 2015 Oct 1. J Cereb Blood Flow Metab. 2016. PMID: 26661172 Free PMC article.
-
Traumatic brain injury from a peripheral axis perspective: Uncovering the roles of liver and adipose tissue in temperature regulation.Prog Neurobiol. 2025 Apr;247:102733. doi: 10.1016/j.pneurobio.2025.102733. Epub 2025 Mar 1. Prog Neurobiol. 2025. PMID: 40032155 Review.
-
Making sense of gut feelings in the traumatic brain injury pathogenesis.Neurosci Biobehav Rev. 2019 Jul;102:345-361. doi: 10.1016/j.neubiorev.2019.05.012. Epub 2019 May 16. Neurosci Biobehav Rev. 2019. PMID: 31102601 Free PMC article. Review.
Cited by
-
Fundamental Neurochemistry Review: Microglial immunometabolism in traumatic brain injury.J Neurochem. 2023 Oct;167(2):129-153. doi: 10.1111/jnc.15959. Epub 2023 Sep 27. J Neurochem. 2023. PMID: 37759406 Free PMC article. Review.
-
Probiotic therapy modulates the brain-gut-liver microbiota axis in a mouse model of traumatic brain injury.Biochim Biophys Acta Mol Basis Dis. 2024 Dec;1870(8):167483. doi: 10.1016/j.bbadis.2024.167483. Epub 2024 Aug 28. Biochim Biophys Acta Mol Basis Dis. 2024. PMID: 39209236 Free PMC article.
-
Traumatic brain injury alters the gut-derived serotonergic system and associated peripheral organs.Biochim Biophys Acta Mol Basis Dis. 2022 Nov 1;1868(11):166491. doi: 10.1016/j.bbadis.2022.166491. Epub 2022 Jul 25. Biochim Biophys Acta Mol Basis Dis. 2022. PMID: 35902006 Free PMC article.
-
Extracranial Effects of Traumatic Brain Injury: A Narrative Review.Clin Pract. 2025 Feb 25;15(3):47. doi: 10.3390/clinpract15030047. Clin Pract. 2025. PMID: 40136583 Free PMC article. Review.
-
The immunological perspective of major depressive disorder: unveiling the interactions between central and peripheral immune mechanisms.J Neuroinflammation. 2025 Jan 19;22(1):10. doi: 10.1186/s12974-024-03312-3. J Neuroinflammation. 2025. PMID: 39828676 Free PMC article. Review.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

