An Emerging Role for the Unfolded Protein Response in Pancreatic Cancer
- PMID: 33445669
- PMCID: PMC7828145
- DOI: 10.3390/cancers13020261
An Emerging Role for the Unfolded Protein Response in Pancreatic Cancer
Abstract
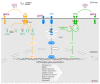
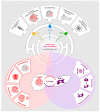
Pancreatic ductal adenocarcinoma (PDAC) is the most common form of pancreatic cancer and one of the leading causes of cancer-associated deaths in the world. It is characterised by dismal response rates to conventional therapies. A major challenge in treatment strategies for PDAC is the presence of a dense stroma that surrounds the tumour cells, shielding them from treatment. This unique tumour microenvironment is fuelled by paracrine signalling between pancreatic cancer cells and supporting stromal cell types including the pancreatic stellate cells (PSC). While our molecular understanding of PDAC is improving, there remains a vital need to develop effective, targeted treatments. The unfolded protein response (UPR) is an elaborate signalling network that governs the cellular response to perturbed protein homeostasis in the endoplasmic reticulum (ER) lumen. There is growing evidence that the UPR is constitutively active in PDAC and may contribute to the disease progression and the acquisition of resistance to therapy. Given the importance of the tumour microenvironment and cytokine signalling in PDAC, and an emerging role for the UPR in shaping the tumour microenvironment and in the regulation of cytokines in other cancer types, this review explores the importance of the UPR in PDAC biology and its potential as a therapeutic target in this disease.
Keywords: activating transcription factor 6 (ATF6); endoplasmic reticulum (ER); inositol-requiring enzyme 1 (IRE1); protein kinase RNA-like ER kinase (PERK); unfolded protein response (UPR).
Conflict of interest statement
A.M.G. and A.S. are co-founders and directors at Cell Stress Discoveries Ltd.
Figures
References
-
- Mayo S.C., Nathan H., Cameron J.L., Olino K., Edil B.H., Herman J.M., Hirose K., Schulick R.D., Choti M.A., Wolfgang C.L., et al. Conditional survival in patients with pancreatic ductal adenocarcinoma resected with curative intent. Cancer. 2011;118:2674–2681. doi: 10.1002/cncr.26553. - DOI - PMC - PubMed
-
- Grossberg A.J., Chu L.C., Deig C.R., Fishman E.K., Hwang W.L., Maitra A., Marks D.L., Mehta A., Nabavizadeh N., Simeone D.M., et al. Multidisciplinary standards of care and recent progress in pancreatic ductal adenocarcinoma. CA Cancer J. Clin. 2020;70:375–403. doi: 10.3322/caac.21626. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

