Premotor Ramping of Thalamic Neuronal Activity Is Modulated by Nigral Inputs and Contributes to Control the Timing of Action Release
- PMID: 33446518
- PMCID: PMC7939094
- DOI: 10.1523/JNEUROSCI.1204-20.2020
Premotor Ramping of Thalamic Neuronal Activity Is Modulated by Nigral Inputs and Contributes to Control the Timing of Action Release
Abstract
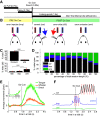
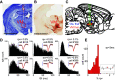
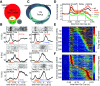
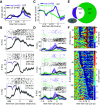
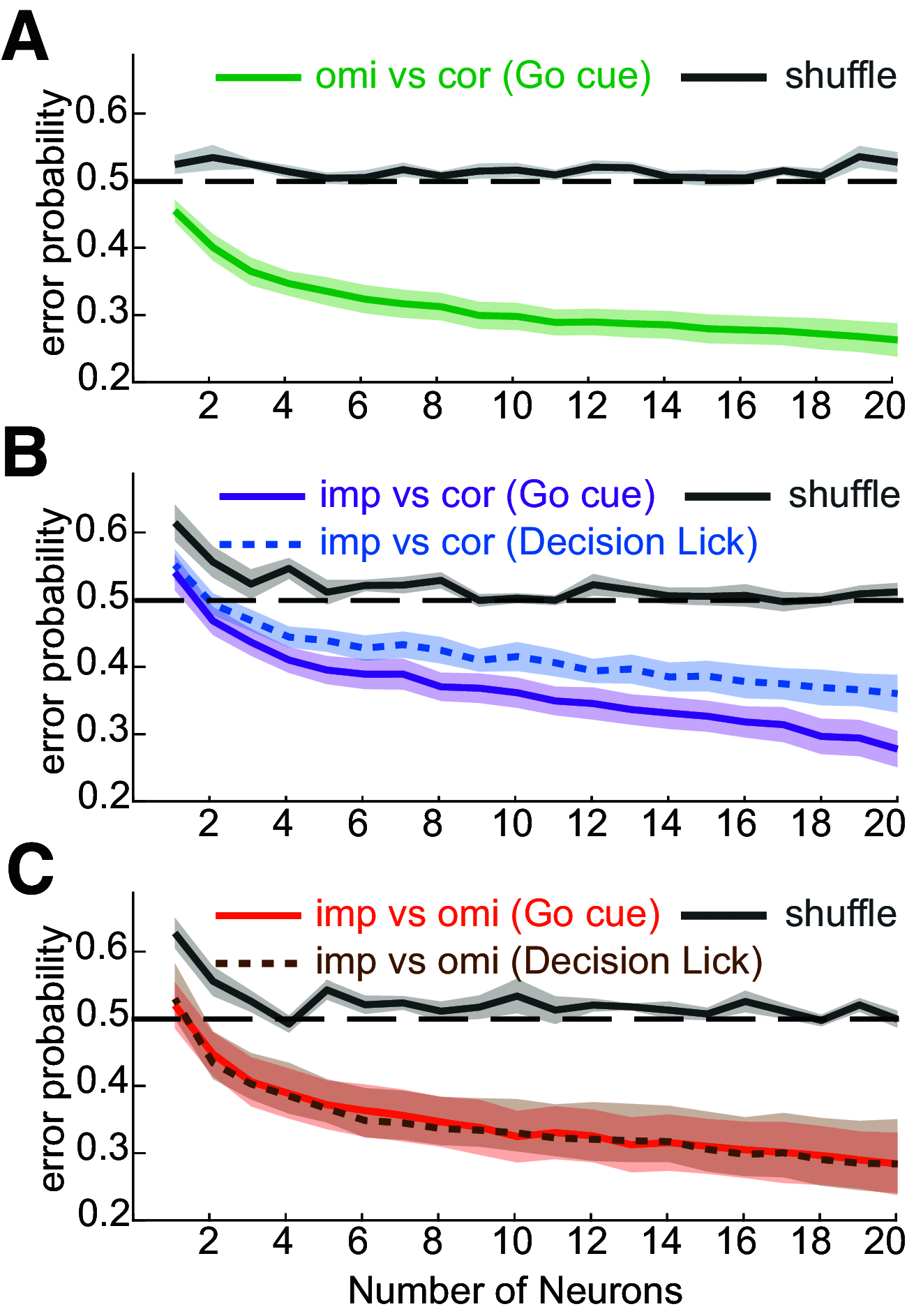
The ventromedial (VM)/ventro-anterior-lateral (VAL) motor thalamus is a key junction within the brain circuits sustaining normal and pathologic motor control functions and decision-making. In this area of thalamus, on one hand, the inhibitory nigro-thalamic pathway provides a main output from the basal ganglia, and, on the other hand, motor thalamo-cortical loops are involved in the maintenance of ramping preparatory activity before goal-directed movements. To better understand the nigral impact on thalamic activity, we recorded electrophysiological responses from VM/VAL neurons while male and female mice were performing a delayed right/left decision licking task. Analysis of correct (corr) and error trials revealed that thalamic ramping activity was stronger for premature licks (impulsive action) and weaker for trials with no licks [omission (omi)] compared with correct trials. Suppressing ramping activity through optogenetic activation of nigral terminals in the motor thalamus during the delay epoch of the task led to a reduced probability of impulsive action and an increased amount of omissions trials. We propose a parsimonious model explaining our data and conclude that a thalamic ramping mechanism contributes to the control of proper timing of action release and that inhibitory nigral inputs are sufficient to interrupt this mechanism and modulate the amount of motor impulsivity in this task.SIGNIFICANCE STATEMENT Coordinated neural activity in motor circuits is essential for correct movement preparation and execution, and even slight imbalances in neural processing can lead to failure in behavioral tasks or motor disorders. Here we focused on how failure to regulate the control of activity balance in the motor thalamus can be implicated in impulsive action release or omissions to act, through an activity ramping mechanism that is required for proper action release. Using optogenetic activation of inhibitory basal ganglia terminals in motor thalamus we show that basal ganglia input is well positioned to control this ramping activity and determine the timing of action initiation.
Keywords: VM thalamus; basal ganglia; behavior; electrophysiology; mouse; optogenetics.
Copyright © 2021 the authors.
Figures
Similar articles
-
Thalamic input to motor cortex facilitates goal-directed action initiation.Curr Biol. 2021 Sep 27;31(18):4148-4155.e4. doi: 10.1016/j.cub.2021.06.089. Epub 2021 Jul 23. Curr Biol. 2021. PMID: 34302741 Free PMC article.
-
Unilateral Optogenetic Inhibition and Excitation of Basal Ganglia Output Affect Directional Lick Choices and Movement Initiation in Mice.Neuroscience. 2019 Dec 15;423:55-65. doi: 10.1016/j.neuroscience.2019.10.031. Epub 2019 Nov 6. Neuroscience. 2019. PMID: 31705892 Free PMC article.
-
Functional anatomy of thalamus and basal ganglia.Childs Nerv Syst. 2002 Aug;18(8):386-404. doi: 10.1007/s00381-002-0604-1. Epub 2002 Jul 26. Childs Nerv Syst. 2002. PMID: 12192499 Review.
-
Anterolateral Motor Cortex Connects with a Medial Subdivision of Ventromedial Thalamus through Cell Type-Specific Circuits, Forming an Excitatory Thalamo-Cortico-Thalamic Loop via Layer 1 Apical Tuft Dendrites of Layer 5B Pyramidal Tract Type Neurons.J Neurosci. 2018 Oct 10;38(41):8787-8797. doi: 10.1523/JNEUROSCI.1333-18.2018. Epub 2018 Aug 24. J Neurosci. 2018. PMID: 30143573 Free PMC article.
-
The functions of the basal ganglia and the paradox of stereotaxic surgery in Parkinson's disease.Brain. 1994 Aug;117 ( Pt 4):877-97. doi: 10.1093/brain/117.4.877. Brain. 1994. PMID: 7922472 Review.
Cited by
-
Thalamus-driven functional populations in frontal cortex support decision-making.Nat Neurosci. 2022 Oct;25(10):1339-1352. doi: 10.1038/s41593-022-01171-w. Epub 2022 Sep 28. Nat Neurosci. 2022. PMID: 36171427 Free PMC article.
-
Input rate encoding and gain control in dendrites of neocortical pyramidal neurons.Cell Rep. 2022 Feb 15;38(7):110382. doi: 10.1016/j.celrep.2022.110382. Cell Rep. 2022. PMID: 35172157 Free PMC article.
-
Tuft dendrites in frontal motor cortex enable flexible learning.bioRxiv [Preprint]. 2025 Mar 13:2025.03.13.642781. doi: 10.1101/2025.03.13.642781. bioRxiv. 2025. PMID: 40161800 Free PMC article. Preprint.
-
Movement-related activity in the internal globus pallidus of the parkinsonian macaque.bioRxiv [Preprint]. 2025 May 30:2024.08.29.610310. doi: 10.1101/2024.08.29.610310. bioRxiv. 2025. Update in: J Neurophysiol. 2025 Aug 1;134(2):741-765. doi: 10.1152/jn.00611.2024. PMID: 39257740 Free PMC article. Updated. Preprint.
-
Movement-related activity in the internal globus pallidus of the parkinsonian macaque.J Neurophysiol. 2025 Aug 1;134(2):741-765. doi: 10.1152/jn.00611.2024. Epub 2025 Jul 22. J Neurophysiol. 2025. PMID: 40695735 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
