Characteristics of monolayer formation in vitro by the chytrid Batrachochytrium dendrobatidis
- PMID: 33447796
- PMCID: PMC7798445
- DOI: 10.1016/j.bioflm.2019.100009
Characteristics of monolayer formation in vitro by the chytrid Batrachochytrium dendrobatidis
Abstract
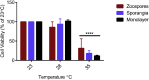
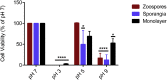
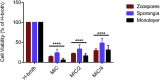
Batrachochytrium dendrobatidis is a globally distributed generalist pathogen that has driven many amphibian populations to extinction. The life cycle of B. dendrobatidis has two main cell types, motile zoospores, and sessile reproductive sporangia. When grown in a nutrient-rich liquid medium, B. dendrobatidis forms aggregates of sporangia that transition into monolayers on surfaces and at the air-liquid interface. Pathogenic microorganisms use biofilms as mechanisms of group interactions to survive under harsh conditions in the absence of a suitable host. We used fluorescent and electron microscopy, crystal violet, transcriptomic, and gas chromatographic analyses to understand the characteristics of B. dendrobatidis monolayers. The cell-free monolayer fraction showed the presence of extracellular ribose, mannose, xylose, galactose, and glucose. Transcriptome analysis showed that 27%, 26%, and 4% of the genes were differentially expressed between sporangia/zoospores, monolayer/zoospores, and sporangia/monolayer pairs respectively. In pond water studies, zoospores developed into sporangia and formed floating aggregates at the air-water interface and attached film on the bottom of growth flasks. We propose that B. dendrobatidis can form surface-attached monolayers in nutrient-rich environments and aggregates of sporangia in nutrient-poor aquatic systems. These monolayers and aggregates may facilitate dispersal and survival of the fungus in the absence of a host. We provide evidence for using a combination of plant-based chemicals, allicin, gingerol, and curcumin as potential anti-chytrid drugs to mitigate chytridiomycosis.
Keywords: Anti-chytrid drugs; Biofilm; Gene expression; Phytochemicals; Transcriptomics.
© 2019 The Authors.
Figures







Similar articles
-
Glutathione-Mediated Metal Tolerance in an Amphibian Chytrid Fungus (Batrachochytrium dendrobatidis).Environ Toxicol Chem. 2024 Jul;43(7):1583-1591. doi: 10.1002/etc.5885. Epub 2024 May 10. Environ Toxicol Chem. 2024. PMID: 38726969
-
Amphibian mucus triggers a developmental transition in the frog-killing chytrid fungus.Curr Biol. 2022 Jun 20;32(12):2765-2771.e4. doi: 10.1016/j.cub.2022.04.006. Epub 2022 Apr 25. Curr Biol. 2022. PMID: 35472310
-
A rapid and inexpensive viability assay for zoospores and zoosporangia of Batrachochytrium dendrobatidis.J Microbiol Methods. 2019 Oct;165:105688. doi: 10.1016/j.mimet.2019.105688. Epub 2019 Aug 16. J Microbiol Methods. 2019. PMID: 31425713
-
Antimicrobial peptide defenses against chytridiomycosis, an emerging infectious disease of amphibian populations.Dev Comp Immunol. 2005;29(7):589-98. doi: 10.1016/j.dci.2004.11.004. Epub 2005 Jan 13. Dev Comp Immunol. 2005. PMID: 15784290 Review.
-
Global emergence of Batrachochytrium dendrobatidis and amphibian chytridiomycosis in space, time, and host.Annu Rev Microbiol. 2009;63:291-310. doi: 10.1146/annurev.micro.091208.073435. Annu Rev Microbiol. 2009. PMID: 19575560 Review.
Cited by
-
Efficacy of Plant-Derived Fungicides at Inhibiting Batrachochytrium salamandrivorans Growth.J Fungi (Basel). 2022 Sep 28;8(10):1025. doi: 10.3390/jof8101025. J Fungi (Basel). 2022. PMID: 36294589 Free PMC article.
-
Frequency-dependent transmission of Batrachochytrium salamandrivorans in eastern newts.Transbound Emerg Dis. 2022 Mar;69(2):731-741. doi: 10.1111/tbed.14043. Epub 2021 Mar 9. Transbound Emerg Dis. 2022. PMID: 33617686 Free PMC article.
-
Amphibian skin bacteria contain a wide repertoire of genes linked to their antifungal capacities.World J Microbiol Biotechnol. 2025 Feb 27;41(3):78. doi: 10.1007/s11274-025-04292-z. World J Microbiol Biotechnol. 2025. PMID: 40011297 Free PMC article.
References
-
- Longcore J.E., Pessier A.P., Nichols D.K. Batrachochytrium dendrobatidis gen. et sp. nov., a chytrid pathogenic to amphibians. Mycologia. 1999;91:219. doi: 10.2307/3761366. - DOI
-
- Yu Y.-H., Zhang S.-C., Zhao Y.-J., Shi J., Yu L.-H., Lu Q.-F. Chytridiomycosis and amphibian population declines. CHINESE J Zool. 2006;41:118–122.
-
- Borovsky D., Smith E.E., Whelan W.J. Purification and properties of potato 1,4-alpha-D-glucan:1,4-alpha-D-glucan 6-alpha-(1,4-alpha-glucano)-transferase. Evidence against a dual catalytic function in amylose-branching enzyme. Eur J Biochem. 1975;59:615–625. doi: 10.1007/s10393-007-0093-5. - DOI - PubMed
LinkOut - more resources
Full Text Sources

