(Macro)molecular self-assembly for hydrogel drug delivery
- PMID: 33450330
- PMCID: PMC8107146
- DOI: 10.1016/j.addr.2021.01.006
(Macro)molecular self-assembly for hydrogel drug delivery
Abstract
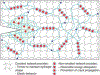
Hydrogels prepared via self-assembly offer scalable and tunable platforms for drug delivery applications. Molecular-scale self-assembly leverages an interplay of attractive and repulsive forces; drugs and other active molecules can be incorporated into such materials by partitioning in hydrophobic domains, affinity-mediated binding, or covalent integration. Peptides have been widely used as building blocks for self-assembly due to facile synthesis, ease of modification with bioactive molecules, and precise molecular-scale control over material properties through tunable interactions. Additional opportunities are manifest in stimuli-responsive self-assembly for more precise drug action. Hydrogels can likewise be fabricated from macromolecular self-assembly, with both synthetic polymers and biopolymers used to prepare materials with controlled mechanical properties and tunable drug release. These include clinical approaches for solubilization and delivery of hydrophobic drugs. To further enhance mechanical properties of hydrogels prepared through self-assembly, recent work has integrated self-assembly motifs with polymeric networks. For example, double-network hydrogels capture the beneficial properties of both self-assembled and covalent networks. The expanding ability to fabricate complex and precise materials, coupled with an improved understanding of biology, will lead to new classes of hydrogels specifically tailored for drug delivery applications.
Keywords: Biomaterials; Block copolymers; Molecular engineering; Peptide self-assembly; Supramolecular chemistry.
Copyright © 2021. Published by Elsevier B.V.
Figures








References
-
- Drury JL and Mooney DJ, Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials, 2003. 24(24): p. 4337–51. - PubMed
-
- Peppas NA, et al., Hydrogels in biology and medicine: From molecular principles to bionanotechnology. Advanced Materials, 2006. 18(11): p. 1345–1360.
-
- Gutowska A, et al., Squeezing hydrogels for controlled oral drug delivery. Journal of Controlled Release, 1997. 48(2-3): p. 141–148.
-
- Jeong B, Bae YH, and Kim SW, Drug release from biodegradable injectable thermosensitive hydrogel of PEG-PLGA-PEG triblock copolymers. Journal of Controlled Release, 2000. 63(1-2): p. 155–163. - PubMed
-
- Nicolson PC and Vogt J, Soft contact lens polymers: an evolution. Biomaterials, 2001. 22(24): p. 3273–3283. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

