Synthesis, inverse docking-assisted identification and in vitro biological characterization of Flavonol-based analogs of fisetin as c-Kit, CDK2 and mTOR inhibitors against melanoma and non-melanoma skin cancers
- PMID: 33450548
- PMCID: PMC7870562
- DOI: 10.1016/j.bioorg.2020.104595
Synthesis, inverse docking-assisted identification and in vitro biological characterization of Flavonol-based analogs of fisetin as c-Kit, CDK2 and mTOR inhibitors against melanoma and non-melanoma skin cancers
Abstract
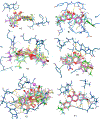
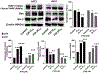
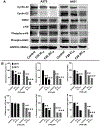
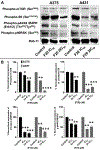
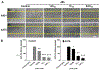
Due to hurdles, including resistance, adverse effects, and poor bioavailability, among others linked with existing therapies, there is an urgent unmet need to devise new, safe, and more effective treatment modalities for skin cancers. Herein, a series of flavonol-based derivatives of fisetin, a plant-based flavonoid identified as an anti-tumorigenic agent targeting the mammalian targets of rapamycin (mTOR)-regulated pathways, were synthesized and fully characterized. New potential inhibitors of receptor tyrosine kinases (c-KITs), cyclin-dependent kinase-2 (CDK2), and mTOR, representing attractive therapeutic targets for melanoma and non-melanoma skin cancers (NMSCs) treatment, were identified using inverse-docking, in vitro kinase activity and various cell-based anticancer screening assays. Eleven compounds exhibited significant inhibitory activities greater than the parent molecule against four human skin cancer cell lines, including melanoma (A375 and SK-Mel-28) and NMSCs (A431 and UWBCC1), with IC50 values ranging from 0.12 to < 15 μM. Seven compounds were identified as potentially potent single, dual or multi-kinase c-KITs, CDK2, and mTOR kinase inhibitors after inverse-docking and screening against twelve known cancer targets, followed by kinase activity profiling. Moreover, the potent compound F20, and the multi-kinase F9 and F17 targeted compounds, markedly decreased scratch wound closure, colony formation, and heightened expression levels of key cancer-promoting pathway molecular targets c-Kit, CDK2, and mTOR. In addition, these compounds downregulated Bcl-2 levels and upregulated Bax and cleaved caspase-3/7/8 and PARP levels, thus inducing apoptosis of A375 and A431 cells in a dose-dependent manner. Overall, compounds F20, F9 and F17, were identified as promising c-Kit, CDK2 and mTOR inhibitors, worthy of further investigation as therapeutics, or as adjuvants to standard therapies for the control of melanoma and NMSCs.
Keywords: Anticancer activity; Apoptosis; Fisetin-analogs; Flavonols; Inverse docking; Kinase activity; Melanoma; Non-melanoma skin cancer.
Published by Elsevier Inc.
Conflict of interest statement
Conflict of interest
The authors declare no conflict of interest.
Figures




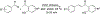
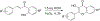
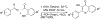
References
-
- Singh PK. Histone methyl transferases: A class of epigenetic opportunities to counter uncontrolled cell proliferation. Eur J Med Chem. 2019,166:351–368. - PubMed
-
- Sun Y Tumor microenvironment and cancer therapy resistance. Cancer Lett. 2016, 380(1):205–215. - PubMed
-
- Santomasso BD. Anticancer drugs and the nervous system. Continuum (Minneap Minn). 2020,26(3):732–764. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

