Missing clinical trial data: the evidence gap in primary data for potential COVID-19 drugs
- PMID: 33451350
- PMCID: PMC7809643
- DOI: 10.1186/s13063-021-05024-y
Missing clinical trial data: the evidence gap in primary data for potential COVID-19 drugs
Abstract
Background: Several drugs are being repurposed for the treatment of the coronavirus disease 2019 (COVID-19) pandemic based on in vitro or early clinical findings. As these drugs are being used in varied regimens and dosages, it is important to enable synthesis of existing safety data from clinical trials. However, availability of safety information is limited by a lack of timely reporting of overall clinical trial results on public registries or through academic publication. We aimed to analyse the evidence gap in this data by conducting a rapid review of results posting on ClinicalTrials.gov and in academic publications to quantify the number of trials missing results for drugs potentially being repurposed for COVID-19.
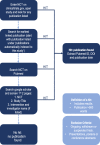
Methods: ClinicalTrials.gov was searched for 19 drugs that have been identified as potential treatments for COVID-19. Relevant clinical trials for any prior indication were listed by identifier (NCT number) and checked for results and for timely result reporting (within 395 days of the primary completion date). Additionally, PubMed and Google Scholar were searched to identify publications of results not listed on the registry. A second, blinded search of 10% of trials was conducted to assess reviewer concordance.
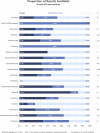
Results: Of 3754 completed trials, 1516 (40.4%) did not post results on ClinicalTrials.gov or in the academic literature. Tabular results were available on ClinicalTrials.gov for 1172 (31.2%) completed trials. A further 1066 (28.4%) had published results in the academic literature, but did not report results on ClinicalTrials.gov . Key drugs missing clinical trial results include hydroxychloroquine (37.0% completed trials unreported), favipiravir (77.8%) and lopinavir (40.5%).
Conclusions: There is an important evidence gap for the safety of drugs being repurposed for COVID-19. This uncertainty could cause unnecessary additional morbidity and mortality during the pandemic. We recommend caution in experimental drug use for non-severe disease and urge clinical trial sponsors to report missing results retrospectively.
Keywords: Adverse events; COVID-19 treatment; Clinical trial transparency; Coronavirus; Repurposed drugs; Safety information.
Conflict of interest statement
Three of the co-authors on this paper are part of Universities Allied for Essential Medicines U.K. However, views expressed in this paper are not necessarily that of Universities Allied for Essential Medicines Europe.
Figures

References
-
- COVID-19 Map - Johns Hopkins Coronavirus Resource Center.
-
- Kalil AC. Treating COVID-19 - off-label drug use, compassionate use, and randomized clinical trials during pandemics. JAMA. 2020;323(19):1897–8. 10.1001/jama.2020.4742. - PubMed
-
- Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for Coronavirus Disease 2019 (COVID-19): a review. JAMA. 2020;323(18):1824–36. 10.1001/jama.2020.6019. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

