Traumatic Brain Injury Causes Chronic Cortical Inflammation and Neuronal Dysfunction Mediated by Microglia
- PMID: 33452227
- PMCID: PMC7896020
- DOI: 10.1523/JNEUROSCI.2469-20.2020
Traumatic Brain Injury Causes Chronic Cortical Inflammation and Neuronal Dysfunction Mediated by Microglia
Abstract
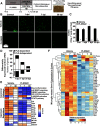
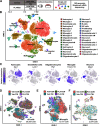
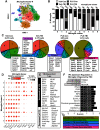
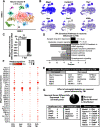
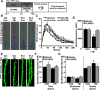
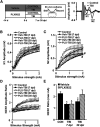
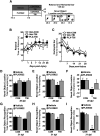
Traumatic brain injury (TBI) can lead to significant neuropsychiatric problems and neurodegenerative pathologies, which develop and persist years after injury. Neuroinflammatory processes evolve over this same period. Therefore, we aimed to determine the contribution of microglia to neuropathology at acute [1 d postinjury (dpi)], subacute (7 dpi), and chronic (30 dpi) time points. Microglia were depleted with PLX5622, a CSF1R antagonist, before midline fluid percussion injury (FPI) in male mice and cortical neuropathology/inflammation was assessed using a neuropathology mRNA panel. Gene expression associated with inflammation and neuropathology were robustly increased acutely after injury (1 dpi) and the majority of this expression was microglia independent. At 7 and 30 dpi, however, microglial depletion reversed TBI-related expression of genes associated with inflammation, interferon signaling, and neuropathology. Myriad suppressed genes at subacute and chronic endpoints were attributed to neurons. To understand the relationship between microglia, neurons, and other glia, single-cell RNA sequencing was completed 7 dpi, a critical time point in the evolution from acute to chronic pathogenesis. Cortical microglia exhibited distinct TBI-associated clustering with increased type-1 interferon and neurodegenerative/damage-related genes. In cortical neurons, genes associated with dopamine signaling, long-term potentiation, calcium signaling, and synaptogenesis were suppressed. Microglial depletion reversed the majority of these neuronal alterations. Furthermore, there was reduced cortical dendritic complexity 7 dpi, reduced neuronal connectively 30 dpi, and cognitive impairment 30 dpi. All of these TBI-associated functional and behavioral impairments were prevented by microglial depletion. Collectively, these studies indicate that microglia promote persistent neuropathology and long-term functional impairments in neuronal homeostasis after TBI.SIGNIFICANCE STATEMENT Millions of traumatic brain injuries (TBIs) occur in the United States alone each year. Survivors face elevated rates of cognitive and psychiatric complications long after the inciting injury. Recent studies of human brain injury link chronic neuroinflammation to adverse neurologic outcomes, suggesting that evolving inflammatory processes may be an opportunity for intervention. Here, we eliminate microglia to compare the effects of diffuse TBI on neurons in the presence and absence of microglia and microglia-mediated inflammation. In the absence of microglia, neurons do not undergo TBI-induced changes in gene transcription or structure. Microglial elimination prevented TBI-induced cognitive changes 30 d postinjury (dpi). Therefore, microglia have a critical role in disrupting neuronal homeostasis after TBI, particularly at subacute and chronic timepoints.
Keywords: CSF1R antagonist; microglia; neuroinflammation; neurotrauma; traumatic brain injury.
Copyright © 2021 the authors.
Figures



References
-
- Bachstetter AD, Webster SJ, Goulding DS, Morton JE, Watterson DM, Van Eldik LJ (2015) Attenuation of traumatic brain injury-induced cognitive impairment in mice by targeting increased cytokine levels with a small molecule experimental therapeutic. J Neuroinflammation 12:69. 10.1186/s12974-015-0289-5 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous
