Regulation of mitochondrial dynamics in 2-methoxyestradiol-mediated osteosarcoma cell death
- PMID: 33452331
- PMCID: PMC7811003
- DOI: 10.1038/s41598-020-80816-x
Regulation of mitochondrial dynamics in 2-methoxyestradiol-mediated osteosarcoma cell death
Abstract
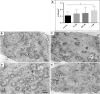
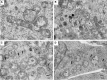
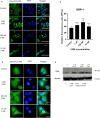
Osteosarcoma (OS) is one of the most malignant tumors of childhood and adolescence. Research on mitochondrial dynamics (fusion/fission) and biogenesis has received much attention in last few years, as they are crucial for death of cancer cells. Specifically, it was shown that increased expression of the cytoplasmic dynamin-related protein 1 (Drp1) triggers mitochondrial fission (division), which activates BAX and downstream intrinsic apoptosis, effectively inhibiting OS growth. In the presented study, human OS cells (metastatic 143B OS cell line) were incubated with 2-methoxyestradiol (2-ME) at both physiologically and pharmacologically relevant concentrations. Cell viability was determined by the MTT assay. Confocal microscopy and western blot methods were applied to examine changes in Drp1 and BAX protein levels. Mitochondrial Division Inhibitor 1, MDIVI-1, was used in the study to further examine the role of Drp1 in 2-ME-mediated mechanism of action. To determine quantitative and qualitative changes in mitochondria, electron microscopy was used. 2-ME at all used concentrations increased mitochondrial fission and induced autophagy in OS cells. At the concentration of 1 µM 2-ME increased the area density of mitochondria in OS cells. Subsequent, upregulated expression of Drp1 and BAX proteins by 2-ME strongly suggests the activation of the intrinsic apoptosis pathway. We further observed 2-ME-mediated regulation of glycolytic state of OS cells. Therefore, we suggest that changes of mitochondrial dynamics may represent a novel mechanism of anticancer action of 2-ME. This finding may open new approaches to improve the efficacy of chemotherapy in the treatment of OS, however, it has to be confirmed by in vivo studies.
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Ciruzzi MS. To mdivi-1 or not to mdivi-1: is that the question? Arch. Argent. Pediatr. 2018;116:365–367. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

