COVID-19 after kidney transplantation: Early outcomes and renal function following antiviral treatment
- PMID: 33453396
- PMCID: PMC7836972
- DOI: 10.1016/j.ijid.2021.01.023
COVID-19 after kidney transplantation: Early outcomes and renal function following antiviral treatment
Abstract
Objectives: The lack of effective treatments for coronavirus disease 2019 (COVID-19) has mandated the repurposing of several drugs, including antiretrovirals and remdesivir (RDV). These compounds may induce acute kidney injury and are not recommended in patients with poor renal function, such as kidney transplant (KTx) recipients.
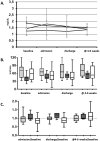
Methods: The records of 42 KTx recipients with COVID-19 were reviewed. Some of them were receiving antiretrovirals (n = 10) or RDV (n = 8) as part of COVID-19 management. Most patients were male (71%) and their median age was 52 years. The median glomerular filtration rate in these patients was 56 ml/min. Regarding disease severity, 36% had mild disease, 19% had moderate disease, 31% had severe disease, and 12% had critical disease. Subgroups, i.e., patients receiving antiretrovirals, RDV, or no antivirals, were comparable in terms of patient age, comorbidities, and immunosuppression.
Results: Seven patients (16.6%) died during hospitalization. Acute kidney injury was found in 24% of KTx recipients at admission. Upon discharge, estimated glomerular filtration rate (eGFR) increased in 32% and decreased in 39% of the KTx recipients compared with the admission rate. The decrease was more prevalent in the RDV group (80%) compared with KTx recipients without any antiviral treatment (29%) (p < 0.05). Most patients (62%) returned to baseline eGFR values within 1 month of discharge. The proportion was similar between the patients receiving antiviral treatment and those not receiving this treatment.
Conclusions: KTx recipients run a high risk of COVID-19-related renal impairment. Antivirals appear to be safe for use without major risks for kidney injury.
Keywords: Antiretrovirals; Complications; Kidney injury; Kidney transplantation; Treatment.
Copyright © 2021 The Author(s). Published by Elsevier Ltd.. All rights reserved.
Figures
References
-
- Alberici F., Delbarba E., Manenti C., Econimo L., Valerio F., Pola A., Maffei C., Possenti S., Zambetti N., Moscato M., Venturini M., Affatato S., Gaggiotti M., Bossini N., Scolari F. A single center observational study of the clinical characteristics and short-term outcome of 20 kidney transplant patients admitted for SARS-CoV2 pneumonia. Kidney Int. 2020;97(6):1083–1088. doi: 10.1016/j.kint.2020.04.002. Jun. - DOI - PMC - PubMed
-
- Argenziano M.G., Bruce S.L., Slater C.L., Tiao J.R., Baldwin M.R., Barr R.G., Chang B.P., Chau K.H., Choi J.J., Gavin N., Goyal P., Mills A.M., Patel A.A., Romney M.S., Safford M.M., Schluger N.W., Sengupta S., Sobieszczyk M.E., Zucker J.E., Asadourian P.A., Bell F.M., Boyd R., Cohen M.F., Colquhoun M.I., Colville L.A., de Jonge J.H., Dershowitz L.B., Dey S.A., Eiseman K.A., Girvin Z.P., Goni D.T., Harb A.A., Herzik N., Householder S., Karaaslan L.E., Lee H., Lieberman E., Ling A., Lu R., Shou A.Y., Sisti A.C., Snow Z.E., Sperring C.P., Xiong Y., Zhou H.W., Natarajan K., Hripcsak G., Chen R. Characterization and clinical course of 1000 patients with coronavirus disease 2019 in New York: retrospective case series. BMJ. 2020;369:m1996. doi: 10.1136/bmj.m1996. May 29. - DOI - PMC - PubMed
-
- Beigel J.H., Tomashek K.M., Dodd L.E., Mehta A.K., Zingman B.S., Kalil A.C., Hohmann E., Chu H.Y., Luetkemeyer A., Kline S., Lopez de Castilla D., Finberg R.W., Dierberg K., Tapson V., Hsieh L., Patterson T.F., Paredes R., Sweeney D.A., Short W.R., Touloumi G., Lye D.C., Ohmagari N., Oh M.D., Ruiz-Palacios G.M., Benfield T., Fätkenheuer G., Kortepeter M.G., Atmar R.L., Creech C.B., Lundgren J., Babiker A.G., Pett S., Neaton J.D., Burgess T.H., Bonnett T., Green M., Makowski M., Osinusi A., Nayak S., Lane H.C. ACTT-1 Study Group Members. Remdesivir for the Treatment of Covid-19 - Final Report. N Engl J Med. 2020;383(19):1813–1826. doi: 10.1056/NEJMoa2007764. Nov 5. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

