Injury measurements improve interpretation of thrombus formation data in the cremaster arteriole laser-induced injury model of thrombosis
- PMID: 33456401
- PMCID: PMC7805486
- DOI: 10.1111/jth.15059
Injury measurements improve interpretation of thrombus formation data in the cremaster arteriole laser-induced injury model of thrombosis
Abstract
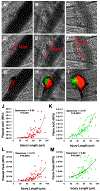
Background: The cremaster arteriole laser-induced injury model is a powerful technique with which to investigate the molecular mechanisms that drive thrombus formation. This model is capable of direct visualization and quantification of accumulation of thrombus constituents, including both platelets and fibrin. However, a large degree of variability in platelet accumulation and fibrin formation is observed between thrombi. Strategies to understand this variability will enhance performance and standardization of the model. We determined whether ablation injury size contributes to variation in platelet accumulation and fibrin formation and, if so, whether incorporating ablation injury size into measurements reduces variation.
Methods: Thrombus formation was initiated by laser-induced injury of cremaster arterioles of mice (n=59 injuries). Ablation injuries within the vessel wall were consistently identified and quantified by measuring the length of vessel wall injury observed immediately following laser-induced disruption. Platelet accumulation and fibrin formation as detected by fluorescently-labeled antibodies were captured by digital intra-vital microscopy.
Results: Laser-induced disruption of the vessel wall resulted in ablation injuries of variable length (18-95 μm) enabling interrogation of the relationship between injury severity and thrombus dynamics. Strong positive correlations were observed between vessel injury length and both platelet and fibrin when the data are transformed as area under the curve (Spearman r = 0.80 and 0.76 respectively). Normalization of area under the curve measurements by injury length reduced intraclass coefficients of variation among thrombi and improved hypothesis testing when comparing different data sets.
Conclusions: Measurement of vessel wall injury length provides a reliable and robust marker of injury severity. Injury length can effectively normalize measurements of platelet accumulation and fibrin formation improving data interpretation and standardization.
Keywords: Animal models; Blood Platelets; Fibrin; Thrombosis.
Conflict of interest statement
Conflict of interest RF is a founder and consultant for Platelet Diagnostics. The other authors have no conflicts of interest to disclose.
Figures

Comment in
-
Control of data variations in intravital microscopy thrombosis models.J Thromb Haemost. 2020 Nov;18(11):2823-2825. doi: 10.1111/jth.15062. J Thromb Haemost. 2020. PMID: 33463084 No abstract available.
References
-
- Raskob G, Angchaisuksiri P, Blanco A, Buller H, Gallus A, Hunt B, Hylek E, Kakkar A, Konstantinides S, McCumber M, Ozaki Y, Wendelboe A, Weitz J. Thrombosis: a major contributor to the global disease burden. Journal of Thrombosis and Haemostasis. 2014; 12: 1580–90. - PubMed
-
- Jackson SP. Arterial thrombosis--insidious, unpredictable and deadly. Nat Med. 2011; 17: 1423–36. 10.1038/nm.2515. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

